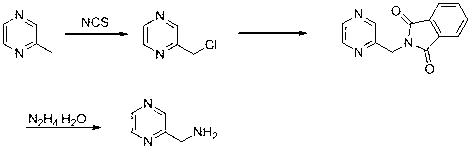
Synthesis method of pyrazine methylamine derivative
A technology of pyrazine methylamine and a synthesis method, which is applied in the field of synthesis of pyrazine methylamine derivatives, can solve the problems of low yield, high cost, complicated steps and the like, and achieves high product yield, few steps and simple operation. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0033] Embodiment 1: the synthesis of 2-chloro-3-methylaminopyrazine
[0034] Add 2-chloro-3-methylpyrazine (75 g, 0.583 mol) and acetonitrile (1050 mL) into a round bottom flask, keep warm in an oil bath at 25 ° C, and add cerium ammonium nitrate (639 g, 1.166 mol) in batches , the addition was completed in about 50 minutes, and the reaction solution was an orange-yellow clear solution. The temperature of the reaction solution was raised to 40°C, and the reaction was carried out for 1 hour. After reaction, the reaction solution was extracted with dichloromethane (650mL×3), the organic phase was dried over anhydrous sodium sulfate, concentrated in vacuo, and rectified (70-75°C, 4mmHg) to obtain 62.3g of light yellow solid (yield 75%) , 2-Chloro-3-formylpyrazine 1 HNMR (CDCl 3 ) δH = 8.62(d,1H), 8.77(d,1H), 10.34(brs,1H).
[0035] Add 2-chloro-3-formylpyrazine (62.3g, 0.437mol) and 10% sodium acetate aqueous solution (500mL, 0.609mol) into a round bottom flask, keep warm in ...
Embodiment 2
[0036] Embodiment 2: the synthesis of 2-methylaminopyrazine
[0037] Add 2-methylpyrazine (27.9g, 0.297mol) and acetic acid (450mL) into a round bottom flask, keep warm in an oil bath at 25°C, add cerium ammonium nitrate (325.5g, 0.594mol) in batches, about 15 After adding within 1 minute, the reaction solution was an orange-yellow clear solution. The reaction solution was heated to 40°C and reacted for 1 hour. At this time, the reaction solution turned dark red. TLC detected that the reaction was complete. Then extract the reaction solution with dichloromethane (350mL×3), dry the organic phase with anhydrous sodium sulfate, concentrate in vacuo, and rectify (52-56°C, 15mmHg) to obtain 26g colorless transparent liquid (yield 81%) , 2-Formylpyrazine 1 HNMR (CDCl 3 ) δH = 8.42-8.76 (m, 1H), 9.20 (s, 1H), 10.17 (brs, 1H).
[0038] Add 2-formylpyrazine (26g, 0.240mol) and 10% sodium acetate aqueous solution (300mL, 0.365mol) into a round-bottomed flask, keep warm in a water bat...
Embodiment 3
[0040] Add 2-methyl-6-carboxypyrazine (5 g, 36.2 mmol, CAS NO.5521-61-9) and water (45 mL), acetic acid (15 ml) into a round-bottomed flask, in an oil bath at 25 °C Keep warm, add cerium ammonium nitrate (39.7g, 72.4mmol) in batches, and add it in about 10 minutes. The reaction solution is an orange-yellow clear solution. The reaction solution is heated to 40°C and reacted for 12 hours. The reaction was detected by TLC, the temperature of the reaction solution was lowered to 10°C, the reaction solution was extracted with dichloromethane (35mL×3), the organic phase was dried with anhydrous sodium sulfate, concentrated in vacuo, and column chromatography (diethyl ether: ethyl acetate = 3: 1) Obtain white solid 2-formyl-6-carboxypyrazine (2.8g, yield 51%)[ 1 HNMR (CDCl 3 ) δH = 8.88 (s,1H), 9.36 (s,H), 10.43 (s,1H), 12.11(brs, 1H)].
[0041]Add 2-formyl-6-carboxylic acid pyrazine (2.8g, 18.4mmol) and pyridine (20ml) into a round bottom flask, keep warm in a water bath at 5°C, a...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More