Recombinant streptomyces diastatochromogenes with reinforced toyF expression, construction method and uses thereof
A technology for producing Streptomyces chromogenes and amylase, which is applied in the field of genetic engineering to achieve the effect of increasing yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] Example 1: Amplification of Target Gene and Construction of Recombinant Amylase Streptomyces Chromogenes
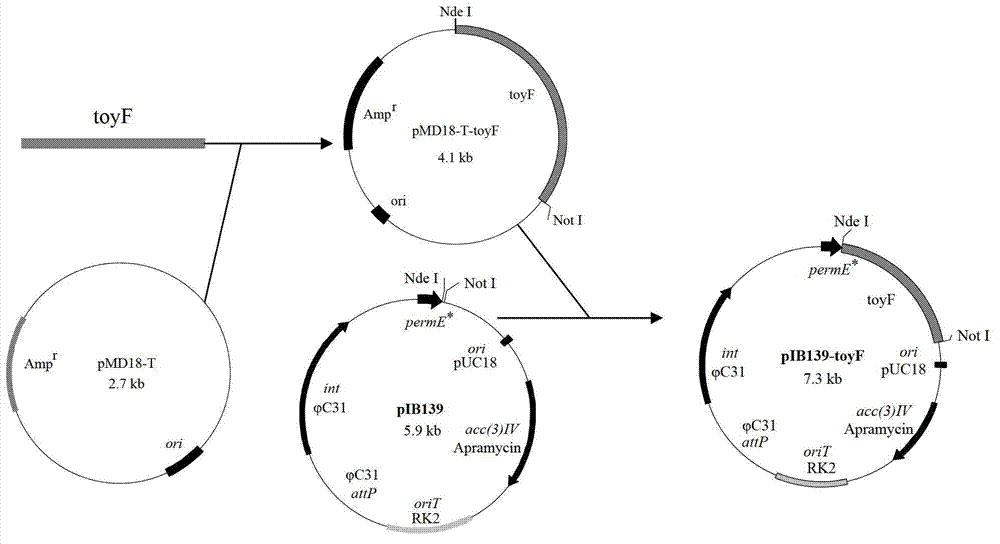
[0030] by S. diastatochromogenes 1628 chromosome genome as template, PtoyF F Nde I and Ptoy F R not I is a primer, PCR amplification obtains containing Nde I and not I The toyF gene with two restriction sites was connected with pMD18-T Vector to construct the cloning vector pMD18-T-toyF ( Nde I + not I), the cloning vector pMD18-T-toyF ( Nde I + not I) Transformed into recipient Escherichia coli, smeared on LB agar plate containing ampicillin resistance, cultured overnight at 37°C, randomly picked positive transformants and sent them to Shanghai Sangong for sequencing and sequence analysis. Nde I and not I double digestion to obtain containing Nde I and not I connect the toyF gene with two restriction sites to the Streptomyces integrated shuttle expression vector pIB139 with the same double digestion, and obtain the recombinant sh...
Embodiment 2
[0036] Example 2: Verification of Fermentation Performance of Amylase Streptomyces Chromogenes Original Strain and Recombinant Strain
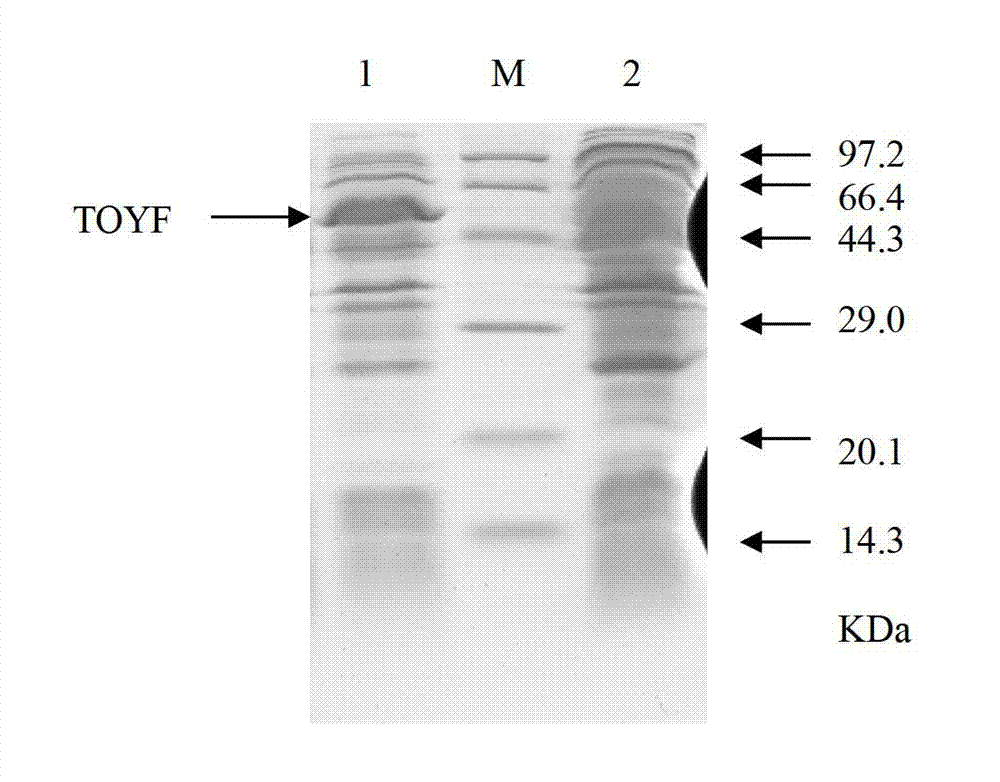
[0037] Compared with the original strain, the adenosine succinate lyase activity of the recombinant strain was increased by 0.9 times, and the recombinant strain 1628-TOYF and the original strain S. diastatochromogenes 1628 conducted a 250 mL shake flask fermentation experiment to verify the effect of the enhancement of adenosuccinate lyase activity on the production of toyocamycin in Streptomyces chromogenes amylase from the perspective of fermentation. The rotating speed of the reciprocating shaker was 200 r / min, 28°C, and fermented for 96 hours. The control group was the origin strain of Streptomyces chromogenes amylase. As shown in Table 1, the yield of the recombinant strain toyocamycin was higher than that of the original strain, and the final yield of the recombinant strain toyocamycin reached 162.64 mg / L, which was about 20.5% high...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Relative molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More