Separation and purification method for (S)-1-amino-3-chloro-2-propanol hydrochloride
A technology of propanol hydrochloride, -1-n-boc-, which is applied in the field of chiral drug intermediates, can solve problems such as the difficulty of debenzylation of hydrochloric acid, achieve the elimination of separation and purification process, improve quality, chemical purity and optical The effect of high purity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
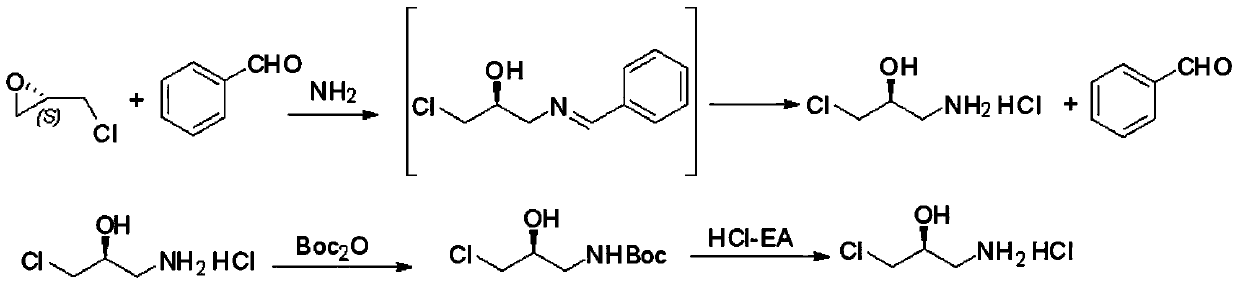
[0026] (1) Synthesis of (S)-1-amino-3-chloro-2-propanol hydrochloride: 270g (5.58mol) 95% ethanol, 106g (1.0mol) benzaldehyde, 100g ( 1.65mol) 28% ammonia water, stirred at 20°C for 1h; added 92.5g (1.0mol) (S)-epichlorohydrin, heated to 40°C for 7h, cooled to 25°C, and kept for 14h to obtain (S)-1 - benzylamino-3-chloro-2-propanol reaction solution; (S)-1-benzylamino-3-chloro-2-propanol reaction solution was added with 300ml of toluene and 300ml of 31% hydrochloric acid, hydrolyzed at 40°C 4h, liquid separation, the water phase was extracted twice with toluene (2×100ml), and the benzaldehyde was separated to obtain (S)-1-amino-3-chloro-2-propanol hydrochloride reaction solution;
[0027] (2) Preparation of (S)-1-N-Boc-amino-3-chloro-2-propanol: (S)-1-amino-3-chloro-2-propanol hydrochloride obtained in step (1) The pH of the salt reaction solution was adjusted to 8 with sodium carbonate, 208 g (1.0 mol) of Boc anhydride was added in batches, and the reaction was carried out a...
Embodiment 2
[0030](1) Synthesis of (S)-1-amino-3-chloro-2-propanol hydrochloride: 340g (7mol) 95% ethanol, 106g (1.0mol) benzaldehyde, 110g (1.8 mol) 28% ammonia water, stirred at 30°C for 1h; added 92.5g (1.0mol) (S)-epichlorohydrin, heated to 50°C for 8h, cooled to 20°C, and kept for 18h to obtain (S)-1- Benzylamino-3-chloro-2-propanol reaction liquid; add 500ml of toluene and 350ml of 31% hydrochloric acid to (S)-1-benzylamino-3-chloro-2-propanol reaction liquid, hydrolyze at 30℃ for 5h , liquid separation, the water phase was extracted twice with toluene (2×100ml), and the benzaldehyde was separated to obtain (S)-1-amino-3-chloro-2-propanol hydrochloride reaction liquid;
[0031] (2) Preparation of (S)-1-N-Boc-amino-3-chloro-2-propanol: (S)-1-amino-3-chloro-2-propanol hydrochloride obtained in step (1) The pH of the salt reaction liquid was adjusted to 9 with sodium carbonate, 250 g (1.2 mol) of Boc anhydride was added in batches, and the reaction was carried out at 20°C for 5 h, and...
Embodiment 3
[0034] (1) Synthesis of (S)-1-amino-3-chloro-2-propanol hydrochloride: 484g (10mol) 95% ethanol, 106g (1.0mol) benzaldehyde, 120g (2.0 mol) 28% ammonia water, stirred at 20°C for 2h; added 92.5g (1.0mol) (S)-epichlorohydrin, heated up to 40°C for 10h, cooled to 30°C, and kept for 10h to obtain (S)-1- Benzylamino-3-chloro-2-propanol reaction liquid; add 400ml of toluene and 400ml of 8mol / l hydrochloric acid to (S)-1-benzylamino-3-chloro-2-propanol reaction liquid, hydrolyze at 50℃ 3h, liquid separation, the water phase was extracted twice with toluene (2×100ml), and the benzaldehyde was separated to obtain (S)-1-amino-3-chloro-2-propanol hydrochloride reaction solution;
[0035] (2) Preparation of (S)-1-N-Boc-amino-3-chloro-2-propanol: (S)-1-amino-3-chloro-2-propanol hydrochloride obtained in step (1) The pH of the salt reaction solution was adjusted to 10 with sodium carbonate, 327 g (1.5 mol) of Boc anhydride was added in batches, and the reaction was carried out at 40°C for...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com