Preparation method for triamcinolone acetonide acetate ultradeformable nanometer lipid vesicle spraying agent and application to target hypertrophic scars
A technology of triamcinolone acetonide acetate and nano-lipids, which is applied in aerosol delivery, medical preparations containing active ingredients, and pharmaceutical formulas. There are no reports on the preparation of vesicle sprays, etc., and the therapeutic effect is remarkable and the effect is long-lasting.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0024] Example 1 Phospholipid ratio screening of triamcinolone acetonide acetate flexible nano lipid vesicle spray
[0025] Prescription group distribution ratio (w / w / %) and dosage:
[0026]
[0027] Prescription two groups distribution ratio (w / w / %) and dosage:
[0028]
[0029]
[0030] Distribution ratio (w / w / %) and dosage of the three prescriptions:
[0031]
[0032] Preparation
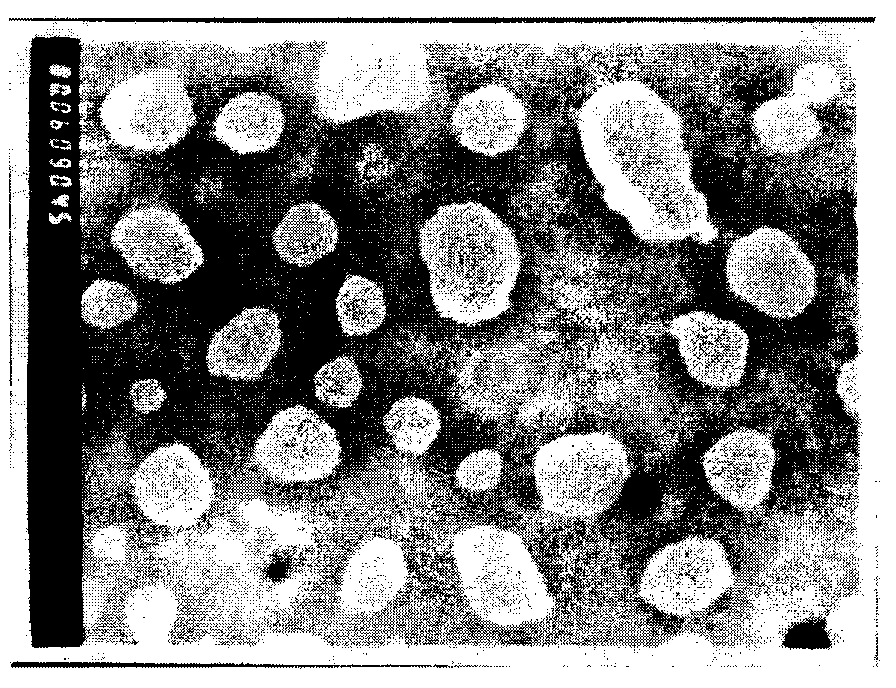
[0033] Quantitatively weigh the phospholipids and triamcinolone acetonide in the above prescriptions into an eggplant-shaped bottle, add 30ml of chloroform-methanol (1:1, v / v) mixed solvent, place in a constant temperature water bath at 37°C, and evaporate the organic solvent under reduced pressure with a water pump , Form a uniform lipid-like film on the inner wall of the bottle, and inject nitrogen gas. Add an aqueous solution of sodium cholate in the prescribed amount, and wash the membrane with rotation for 2 hours to obtain a flexible nano-lipid vesicle suspension. Under th...
Embodiment 2
[0043] Example 2 Sodium cholate ratio screening of triamcinolone acetonide acetate flexible nano lipid vesicle spray
[0044] Prescription group distribution ratio (w / w / %) and dosage:
[0045]
[0046] Prescription two groups distribution ratio (w / w / %) and dosage:
[0047]
[0048] Distribution ratio (w / w / %) and dosage of the three prescriptions:
[0049]
[0050]
[0051] Preparation
[0052] Quantitatively weigh the phospholipids and triamcinolone acetonide in the above prescriptions into an eggplant-shaped bottle, add 30ml of chloroform-methanol (1:1, v / v) mixed solvent, place in a constant temperature water bath at 37°C, and evaporate the organic solvent under reduced pressure with a water pump , Form a uniform lipid-like film on the inner wall of the bottle, and inject nitrogen gas. Add an aqueous solution of sodium cholate in the prescribed amount, and wash the membrane with rotation for 2 hours to obtain a flexible nano-lipid vesicle suspension. Under ...
Embodiment 3
[0065] Embodiment 3 Ex vivo scar skin drug release experiment
[0066] skin preparation
[0067] Patients with hypertrophic scars with a course of more than 1 year, relatively flat local scars, no ulceration, an area of more than 3 cm × 3 cm, and requiring surgical resection were selected, and informed consent was given before surgery. After removing the scarred skin, the subcutaneous fat was removed, rinsed with normal saline, and stored at -20°C for later use.
[0068] Penetration test
[0069] Using the improved Franz diffusion cell, the skin is fixed between the supply pool and the receiving pool, the stratum corneum faces the supply pool, and the effective penetration area is 0.952cm 2 . The supply pools were put into prescription one to prepare 0.1% triamcinolone acetonide acetate flexible nano-lipid vesicle spray, 0.1% triamcinolone acetonide acetate nano-lipid vesicles, 0.1% triamcinolone acetonide solution, commercially available triamcinolone acetonide milk ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| The average particle size | aaaaa | aaaaa |
| The average particle size | aaaaa | aaaaa |
| Particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More