Sarpogrelate hydrochloride sustained release preparation
A technology of sargrelate hydrochloride and sustained-release preparations, which is applied in the field of pharmaceutical preparations, can solve the problems of digestive tract irritation, poor compliance, and inconvenient medication for patients, and achieve the effect of simple preparation process
Inactive Publication Date: 2015-05-06
济南齐拓医药科技有限公司
View PDF5 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
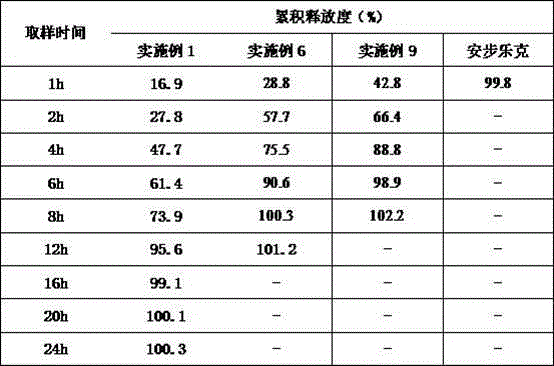
[0006] 1) The in vitro release shows that the drug release reaches more than 80% after 30 minutes, and the emptying time of the stomach is about 2 hours. Therefore, most of the drugs are released in the stomach, and the local concentration is high, so it is easy to cause digestive tract irritation , produce nausea, heartburn, gastrointestinal bleeding and other adverse reactions;
[0007] 2) Sagrelate Hydrochloride Tablets need to be taken 3 times a day, which is inconvenient for patients and poor compliance
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0044] Sagrelate Hydrochloride 50%
[0045] Polyvinyl Acetate Povidone Blend 40%
[0046] Microcrystalline Cellulose 8%
[0047] Povidone K30 1%
Embodiment 2
[0050] Sagrelate Hydrochloride 40%
[0051] Polyvinyl Acetate Povidone Blend 50%
[0052] Microcrystalline Cellulose 8%
[0053] Povidone K30 1%
Embodiment 3
[0056] Sagrelate Hydrochloride 40%
[0057] Polyvinyl Acetate Povidone Blend 40%
[0058] Microcrystalline Cellulose 18%
[0059] Povidone K30 1%
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention belongs to the field of pharmaceutical preparations and discloses a sarpogrelate hydrochloride sustained release preparation. The sustained release preparation consists of sarpogrelate hydrochloride, a polyvinyl acetate povidone mixture and other auxiliary materials.
Description
technical field [0001] The invention relates to the field of pharmaceutical preparations. Background technique [0002] Sarpogrelate Hydrochloride (Sarpogrelate Hydrochloride), molecular formula: C 24 h 31 NO 6 HCl, with a molecular weight of 465.97, is a 5-HT receptor blocker developed by Mitsubishi Corporation of Japan. Its pharmacological effects include the following aspects: it has specific antagonistic effect on the 5-HT2 receptor of platelets and vascular smooth muscle; it inhibits platelet aggregation Effect; Antithrombotic effect; Inhibition of vasoconstriction; Improvement of microcirculation. [0003] Currently the only product on the market is sargrelate hydrochloride tablets, the trade name is Anbu Leke, and the specification is 100mg. It is clinically used to improve ischemic symptoms such as ulcer pain and cold sensation caused by chronic arterial occlusive disease. Usage and dosage: Adults take 100mg three times a day, orally after meals, but the dosage s...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): A61K31/225A61K9/22A61K47/32A61P7/02A61P9/00
Inventor 王栋宋永亮王振沈华亭王润华
Owner 济南齐拓医药科技有限公司