Method for preparing super paramagnetic carrier of cross-linking immobilized lipase
A fat immobilization and superparamagnetic technology, applied in the field of food chemistry, can solve problems such as inability to adapt to lipase interface activation characteristics, functional defects, damage, etc., achieve good magnetic separation performance, and overcome the effect of lipase activity-dependent interface effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
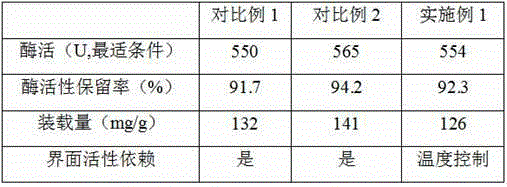
Embodiment 1
[0024] (1) Nano-magnetic microsphere Fe 3 o 4 Preparation of:
[0025] FeCl 3 ·6H 2 O was dissolved in anhydrous ethylene glycol to prepare a solution with a concentration of 0.13mmol / mL, and then press FeCl 3 ·6H 2 The mass ratio of O to the treatment agent is 1:6. Add the treatment agent, wherein the treatment agent is sodium acetate and polyethylene glycol. After adding the treatment agent, the concentration of sodium acetate is 95mg / mL, and then transfer to the polytetrafluoroethylene coated with inert material. In a reactor made of vinyl fluoride, ultrasonically react at a constant temperature of 200°C for 5 hours. After natural cooling, wash with ethanol and deionized water alternately for several times, then wash with deionized water, and then dry in vacuum to obtain nano-magnetic particles. Ball Fe 3 o 4 ;
[0026] (2) Nano-magnetic microsphere Fe3 o 4 The package:
[0027] Nano magnetic microsphere Fe obtained in step (1) 3 o 4 Mix with the NaOH solution w...
Embodiment 2
[0032] (1) Nano-magnetic microsphere Fe 3 o 4 Preparation of:
[0033] FeCl 3 ·6H 2 O was dissolved in anhydrous ethylene glycol to prepare a solution with a concentration of 0.1mmol / mL, and then press FeCl 3 ·6H 2 The mass ratio of O to the treatment agent is 1:5. Add the treatment agent, wherein the treatment agent is sodium acetate and polyethylene glycol. After adding the treatment agent, the concentration of sodium acetate is 120mg / mL, and then transfer to the polytetrafluoroethylene coated with inert material. In a reactor made of vinyl fluoride, ultrasonically react at a constant temperature of 200°C for 6 hours. After natural cooling, first wash with ethanol and deionized water alternately for several times, then wash with deionized water, and then vacuum dry to obtain nano-magnetic particles. Ball Fe 3 o 4 ;
[0034] (2) Nano-magnetic microsphere Fe 3 o 4 The package:
[0035] Nano magnetic microsphere Fe obtained in step (1) 3 o 4 Mix with the NaOH solut...
Embodiment 3
[0040] (1) Nano-magnetic microsphere Fe 3 o 4 Preparation of:
[0041] FeCl 3 ·6H 2 O was dissolved in anhydrous ethylene glycol to prepare a solution with a concentration of 0.15mmol / mL, and then press FeCl 3 ·6H 2 The mass ratio of O to the treatment agent is 1:4. Add the treatment agent, wherein the treatment agent is sodium acetate and polyethylene glycol. After adding the treatment agent, the concentration of sodium acetate is 80 mg / mL, and then transfer to the polytetrafluoroethylene coated with inert material. In the vinyl fluoride reactor, ultrasonically react at a constant temperature of 200°C for 4 hours. After natural cooling, wash with ethanol and deionized water alternately for several times, then wash with deionized water, and then dry in vacuum to obtain nano-magnetic particles. Ball Fe 3 o 4 ;
[0042] (2) Nano-magnetic microsphere Fe 3 o 4 The package:
[0043] Nano magnetic microsphere Fe obtained in step (1) 3 o 4 Mix with the NaOH solution whos...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Concentration | aaaaa | aaaaa |
| Particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More