Medicinal composition for activating blood circulation, dissipating blood stasis, warming meridians and relaxing veins, and preparation method thereof
A technology that warms the meridian, unblocks collaterals, activates blood circulation and removes blood stasis. It is applied in drug combinations, active ingredients of hydroxyl compounds, and pharmaceutical formulas. It can solve problems such as large toxic and side effects, strong dependence, and stimulation of the digestive nervous system, and improve product quality and dissolution. degree of effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
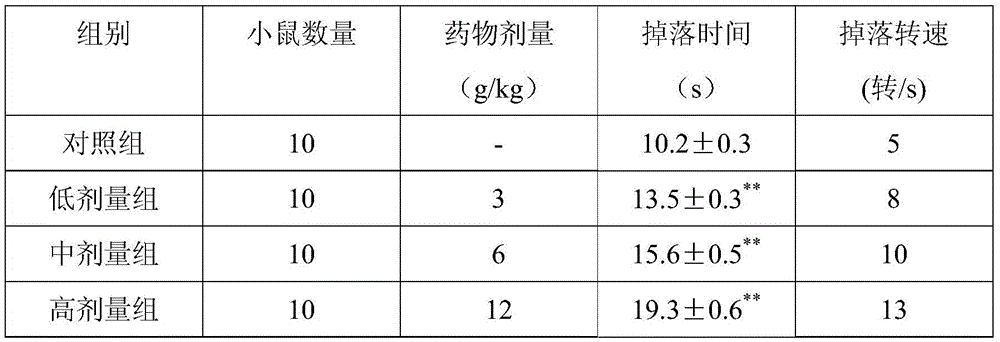
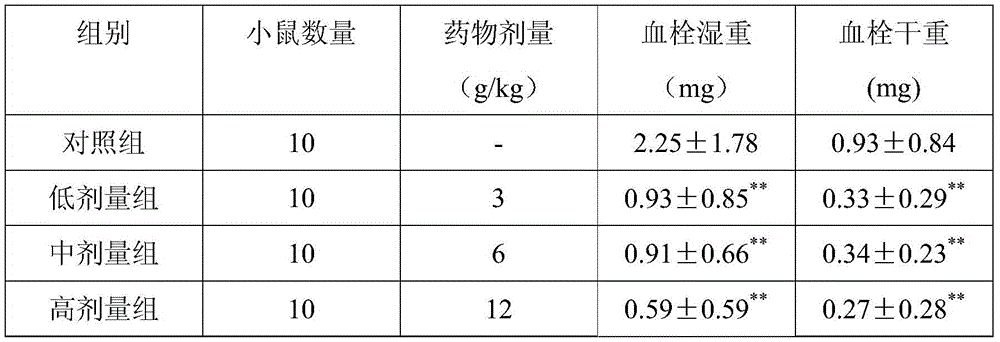
Examples
Embodiment 1
[0028] Made from the following raw materials in parts by weight:
[0029] 260 parts of Chuanxiong, 410 parts of Astragalus, 120 parts of Panax notoginseng, 120 parts of Guizhi, 40 parts of woody fragrance, 120 parts of hawthorn, 200 parts of salvia miltiorrhiza, 120 parts of Alisma, 40 parts of Sophora japonica, 120 parts of turmeric, 5 parts of borneol;
[0030] The preparation method of the active ingredient of the pharmaceutical composition of the present invention comprises the following steps:
[0031] (1) Take Rhizoma Chuanxiong in proportion, add 1% compound enzyme containing cellulase and pectinase, wherein the ratio of cellulase and pectinase is 1:1, add 1 times the amount of warm water, mix well, 40 Enzyme hydrolysis at ℃ for 1 hour; add water, extract once by steam distillation, collect volatile oil, filter the medicinal solution, combine the filtrates, concentrate with a flash evaporator, add a large amount of ethanol and let it stand overnight, centrifuge with a h...
Embodiment 2
[0043] Made from the following raw materials in parts by weight:
[0044] 280 parts of Chuanxiong, 430 parts of Astragalus, 140 parts of Panax notoginseng, 140 parts of Guizhi, 60 parts of woody fragrance, 140 parts of hawthorn, 220 parts of salvia miltiorrhiza, 140 parts of Alisma, 60 parts of Sophora japonica, 140 parts of turmeric, 6.5 parts of borneol.
[0045] The preparation method of the active ingredient of the pharmaceutical composition of the present invention comprises the following steps:
[0046] (1) Take Rhizoma Chuanxiong in proportion, add 5.5% compound enzyme containing cellulase and pectinase, wherein the ratio of cellulase and pectinase is 1:1, add 5.5 times the amount of warm water, mix, 50 Enzyme hydrolysis at ℃ for 3 hours; add water, extract 3 times by steam distillation, collect volatile oil, filter the liquid medicine, combine the filtrates, concentrate with a flash evaporator, add a large amount of ethanol and let it stand overnight, centrifuge with a...
Embodiment 3
[0058] Made from the following raw materials in parts by weight:
[0059] 300 parts of Rhizoma Chuanxiong, 450 parts of Astragalus, 160 parts of Panax notoginseng, 160 parts of Guizhi, 80 parts of woody fragrance, 160 parts of hawthorn, 240 parts of salvia miltiorrhiza, 160 parts of Alisma, 80 parts of Sophora japonica, 160 parts of turmeric, 8 parts of borneol;
[0060] The preparation method of the active ingredient of the pharmaceutical composition of the present invention comprises the following steps:
[0061] (1) Take Rhizoma Chuanxiong in proportion, add 10% compound enzyme containing cellulase and pectinase, wherein the ratio of cellulase and pectinase is 1:1, add 10 times the amount of warm water, mix well, 60 Enzyme hydrolysis at ℃ for 5 hours; add water, use steam distillation to extract 5 times, collect volatile oil, filter the liquid medicine, combine the filtrates, use a flash evaporator to concentrate, add a large amount of ethanol to stand overnight, use a high...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap