Method for simultaneous determination of double items of urea nitrogen and creatinine in serum
A determination method, urea nitrogen technology, applied in biochemical equipment and methods, determination/inspection of microorganisms, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0028] Composition of reagents:
[0029] a. Reagent I:
[0030] Reagent Ⅰ contains TRIS-hydrochloride phosphate buffer 50mmol / L, urease 5000U / L, glutamate dehydrogenase 9000U / L, α-ketoglutarate 160mmol / L, NADH 3.0mmol / L, Proclin-300 150-250 μl.
[0031] b. Reagent II:
[0032] Reagent II contains TRIS-hydrochloride phosphate buffer 50mmol / L, creatinine amidohydrolase 20KU / L, creatine amidinohydrolase 20KU / L, Proclin-300 150-250μl.
[0033] c. Standard solution: The standard solution is 7.14mmol / urea nitrogen.
Embodiment 2
[0035] Measurement procedure
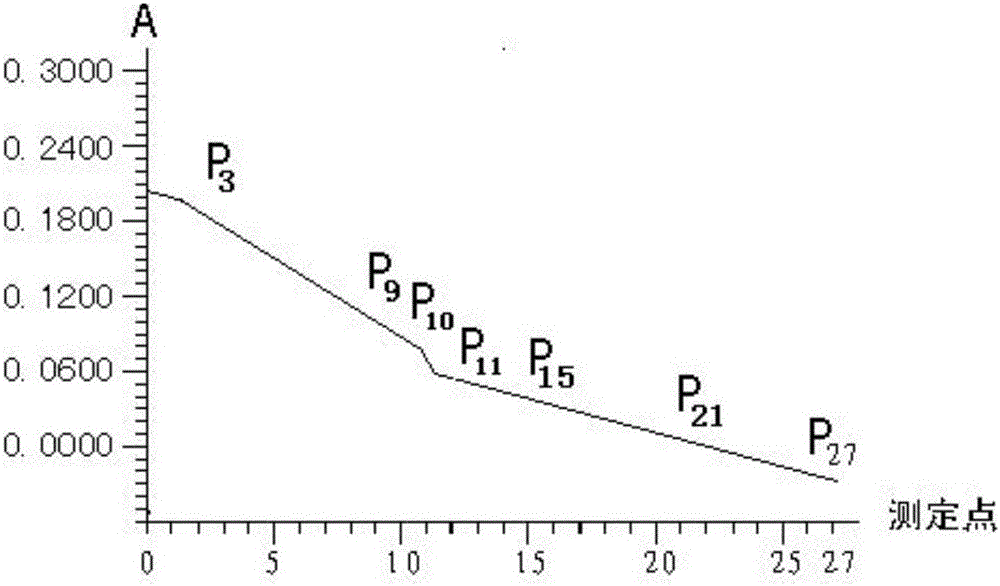
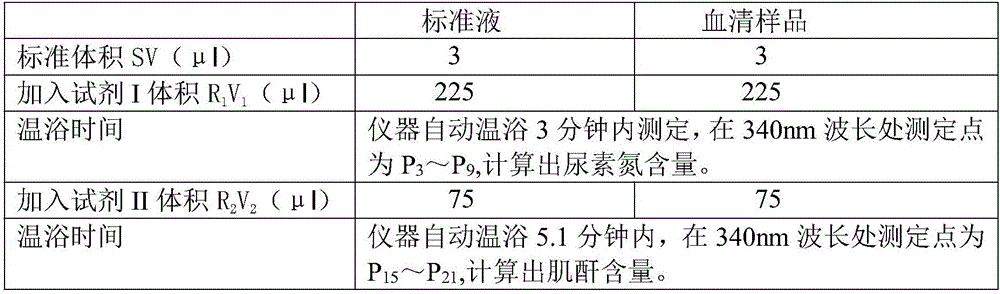
[0036] Determination method: On the Japanese OLYMPUS AU2700 fully automated biochemical analyzer, the instrument automatically adds 3 μl of sample to 225 μl of reagent Ⅰ and mixes well, incubates at 37°C for 3 minutes, adds 75 μl of reagent Ⅱ, mixes well, and incubates at 37°C for 5.1 minutes, fully automatic analysis The instrument detects at a wavelength of 340nm. The instrument automatically calculates the results of urea nitrogen and creatinine, see Table 1 for details:
[0037] Table 1. Automatic biochemical analyzer test conditions of the present invention
[0038]
[0039] Concentration of urea nitrogen = F × ΔOD BUN / Δt
[0040] Creatinine concentration = F × ΔOD Cr / Δt
[0041] where ΔOD BUN / Δt is the rate of urea nitrogen production per unit time, ΔOD Cr / Δt is the rate of creatinine production per unit time, F is the correction factor, the measurement method is the rate method with standard, and its real-time response curve ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More