A kind of preparation method of iomeprol impurity
A compound, methylamino technology, used in the preparation of organic compounds, the preparation of carboxylic acid amides, chemical instruments and methods, etc., can solve problems such as adverse reactions, and achieve the effects of stable quality, mild reaction conditions and high purity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
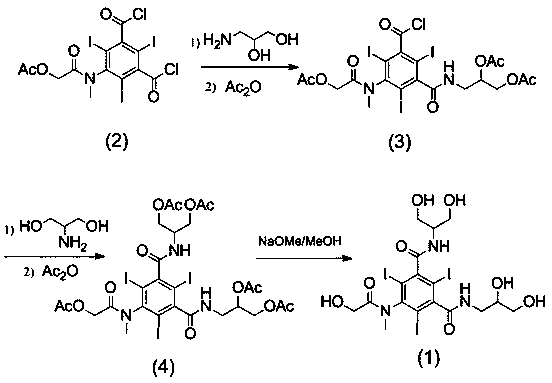
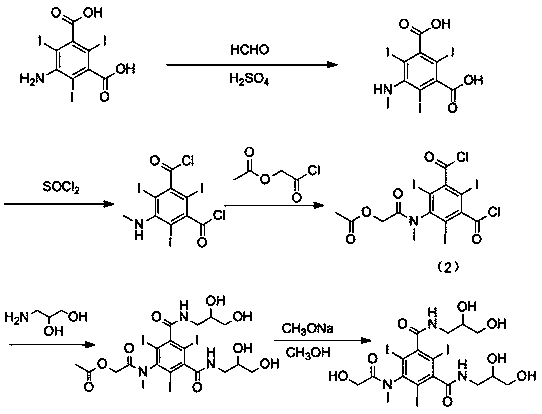
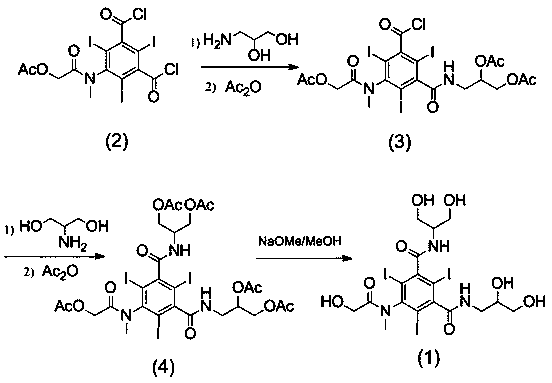
[0029] Example 1: Compound 3-N-(2,3-diacetoxypropylcarbamoyl)-5-[(acetoxyacetyl)methylamino]-2,4,6- Synthesis of Triiodobenzoyl Chloride
[0030]The compound of formula (2) 5-[(acetoxyacetyl)methylamino]-2,4,6-triiodo-1,3-phthaloyl chloride (10g) was dissolved in N,N-dimethyl In acetamide (20ml), dropwise into 3-amino-1,2-propanediol (1.3g, dissolved in 6ml N,N-dimethylacetamide and 2.3ml triethylamine), control the reaction temperature during the dropwise addition The temperature is 0-5°C, and the reaction is kept for 1 hour after dropping. After the reaction is completed, add 4-dimethylaminopyridine (0.1g), dropwise add acetic anhydride (3.4ml), control the temperature and react at less than 30°C, and react at 25-30°C for 2h after dropping. After the reaction is complete, add water (20ml), stir for 10min, then add dichloromethane (45ml), stir for 30min, then separate layers, extract the aqueous layer once with dichloromethane (45ml), combine the organic layers, add water (...
Embodiment 2
[0031] Example 2: Formula (4) compound N-(1,3-diacetoxypropyl)-N ’ -Synthesis of (2,3-diacetoxypropyl)-5-[(acetoxyacetyl)methylamino]-2,4,6-triiodo-1,3-benzenedicarboxamide
[0032] The formula (3) compound 3-N-(2,3-diacetoxypropylcarbamoyl)-5-[(acetoxyacetyl)methylamino]-2,4,6-triiodobenzene Refined formyl chloride (6.5g) was dissolved in N,N-dimethylacetamide (13ml), dropped into 2-amino-1,3-propanediol (dissolved in 7ml N,N-dimethylacetamide and 1.5 ml triethylamine), after dropping, keep the reaction at 20-25°C for 1.5h. After the reaction is complete, add 4-dimethylaminopyridine (0.07g), dropwise add acetic anhydride (2.2ml), control the temperature below 30°C, and react at 25-30°C for 2h after dropping. After the reaction is complete, add water (13ml), stir for 10min, then add dichloromethane (25ml), stir for 30min, then separate layers, extract the aqueous layer once with dichloromethane (25ml), combine the organic layers, add water (13ml) and stir, Adjust the pH to ...
Embodiment 3
[0033] Embodiment 3: Formula (1) compound N-(1,3-dihydroxypropyl)-N ’ -Synthesis of (2,3-dihydroxypropyl)-5-[(hydroxyacetyl)methylamino]-2,4,6-triiodo-1,3-benzenedicarboxamide
[0034] The formula (4) compound N-(1,3-diacetoxypropyl)-N ’ -(2,3-diacetoxypropyl)-5-[(acetoxyacetyl)methylamino]-2,4,6-triiodo-1,3-benzenedicarboxamide (5.7g) Dissolve in methanol (45ml), add 30% sodium methoxide methanol solution dropwise to make the pH 8-9, and react at 25°C for 1h until the reaction is complete. Add cationic resin to make the pH to 7, filter with suction, and evaporate methanol to dryness under reduced pressure to obtain the crude compound of formula (1) (4.3 g, HPLC purity 96%). Dissolve the crude compound of formula (1) in water (20ml), add anionic resin, stir for 0.5h, filter with suction, and evaporate to dryness under reduced pressure to obtain the refined compound of formula (1) (3.7g, HPLC purity greater than 99%). 1 H-NMR (800MHz, DMSO- d 6 ) δ (ppm): 8.45-8.59 (m, 2H...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com