Dextromethorphan hydrobromide orally disintegrating tablet and preparation method thereof
A technology of dextromethorphan hydrobromide and orally disintegrating tablets, which is applied in the field of medicine, can solve problems such as poor compliance, achieve low production cost, short disintegration time, and meet the production requirements
Inactive Publication Date: 2017-03-29
HARBIN KEDI PHARMA +1
View PDF2 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
However, the elderly, children and some patients with swallowing dysfunction have poor compliance with the above preparations
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
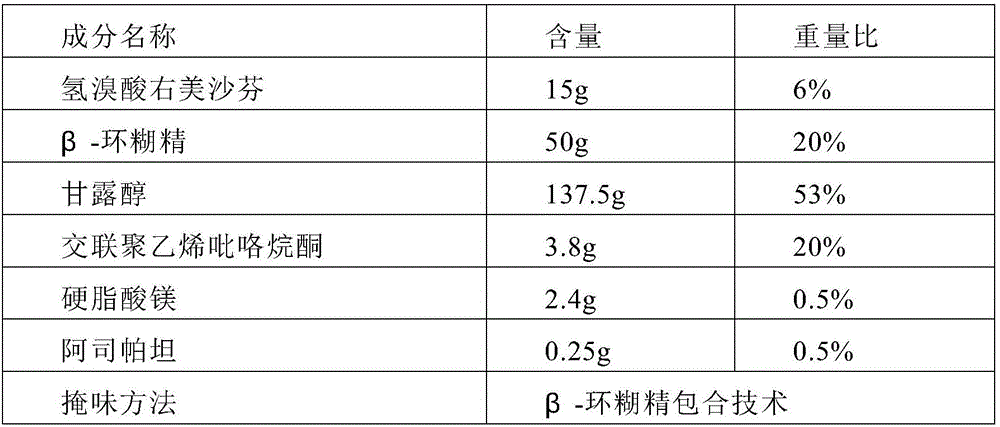
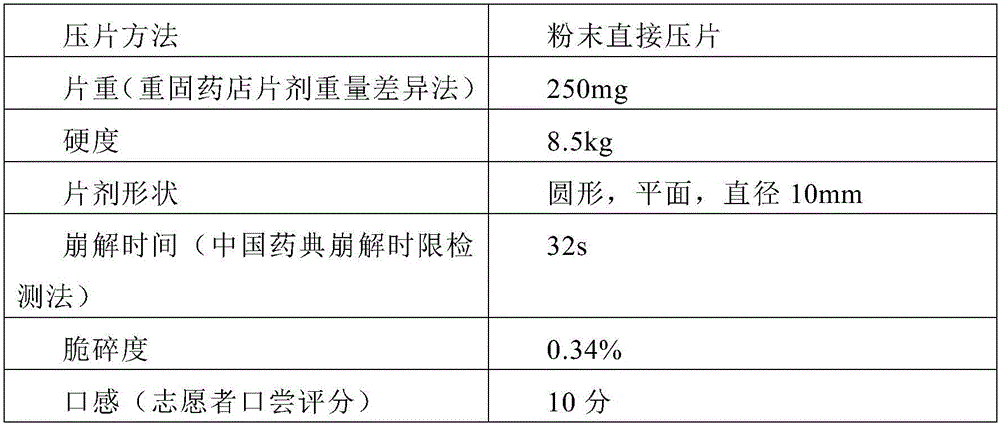
[0047]Prepare 1000 tablets of dextromethorphan hydrobromide orally disintegrating tablets (specification: dextromethorphan hydrobromide 15mg)
[0048]
[0049]
Embodiment 2
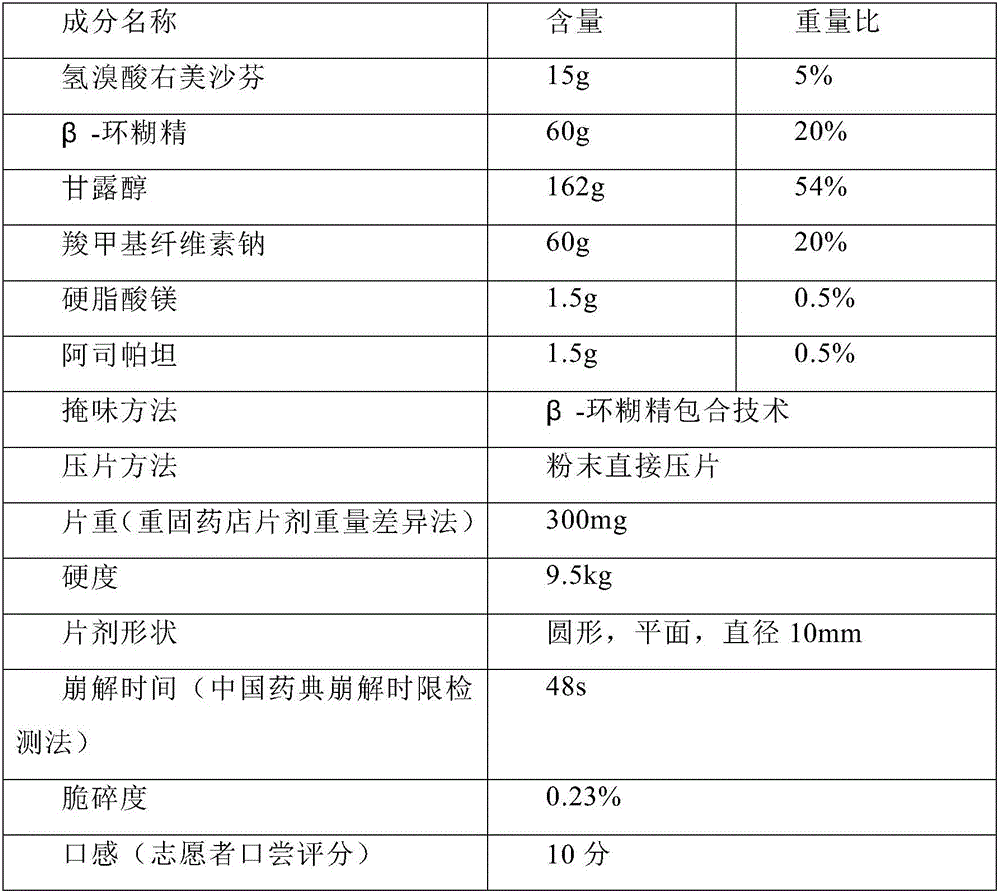
[0051] Prepare 1000 tablets of dextromethorphan hydrobromide orally disintegrating tablets (specification: dextromethorphan hydrobromide 15mg)
[0052]
Embodiment 3
[0054] Prepare 1000 tablets of dextromethorphan hydrobromide orally disintegrating tablets (specification: dextromethorphan hydrobromide 15mg)
[0055]
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention discloses a dextromethorphan hydrobromide orally disintegrating tablet, which comprises dextromethorphan hydrobromide and auxiliary materials. The auxiliary materials are selected from one or more of beta-cyclodextrin, mannitol, microcrystalline cellulose, magnesium stearate, crosslinked polyvinylpyrrolidone, aspartame, sodium carboxymethylcellulose, lactose, starch, glyceryl behenate, crosslinked carboxymethyl starch and lauryl sodium sulfate. Compared with the prior art, the dextromethorphan hydrobromide orally disintegrating tablet provided by the invention has shorter disintegration time and better taste than traditional tablets and capsules, can rapidly disintegrate and dissolve out in an oral cavity without the need for taking with water, takes effect rapidly, and can be conveniently taken by the aged, the children and patients with difficulty in taking medicine, and the effectiveness, the safety and the compliance of a medicine are improved. A preparation method adopts common equipment, and is low in production cost and suitable for industrial production. The dextromethorphan hydrobromide orally disintegrating tablet is applicable to relieving throat discomfort caused by cold, acute tracheitis, bronchitis and allergy.
Description
technical field [0001] The invention relates to a dextromethorphan hydrobromide orally disintegrating tablet and a preparation method thereof, belonging to the technical field of medicine. Background technique [0002] With the increasing air pollution in recent years, the number of patients with respiratory diseases has also increased. The drug use of the elderly, young children and patients with dysphagia is also one of the key points of research by medical workers. In recent years, orally disintegrating tablets, as a new type of oral preparation, have largely solved this problem. [0003] As a new type of oral tablet, orally disintegrating tablet has clear requirements for its disintegration time limit and environment. The Drug Evaluation Center of the State Food and Drug Administration (SFDA) established the basic quality control requirements for the disintegration time limit of orally disintegrating tablets in the "Dosage Form Characteristics and Quality Control Meeti...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): A61K9/20A61K31/485A61K47/61A61P11/14A61P11/04
CPCA61K9/0056A61K9/205A61K31/485
Inventor 吕宝光张文君张国锋徐铭凯李月月王嵩
Owner HARBIN KEDI PHARMA