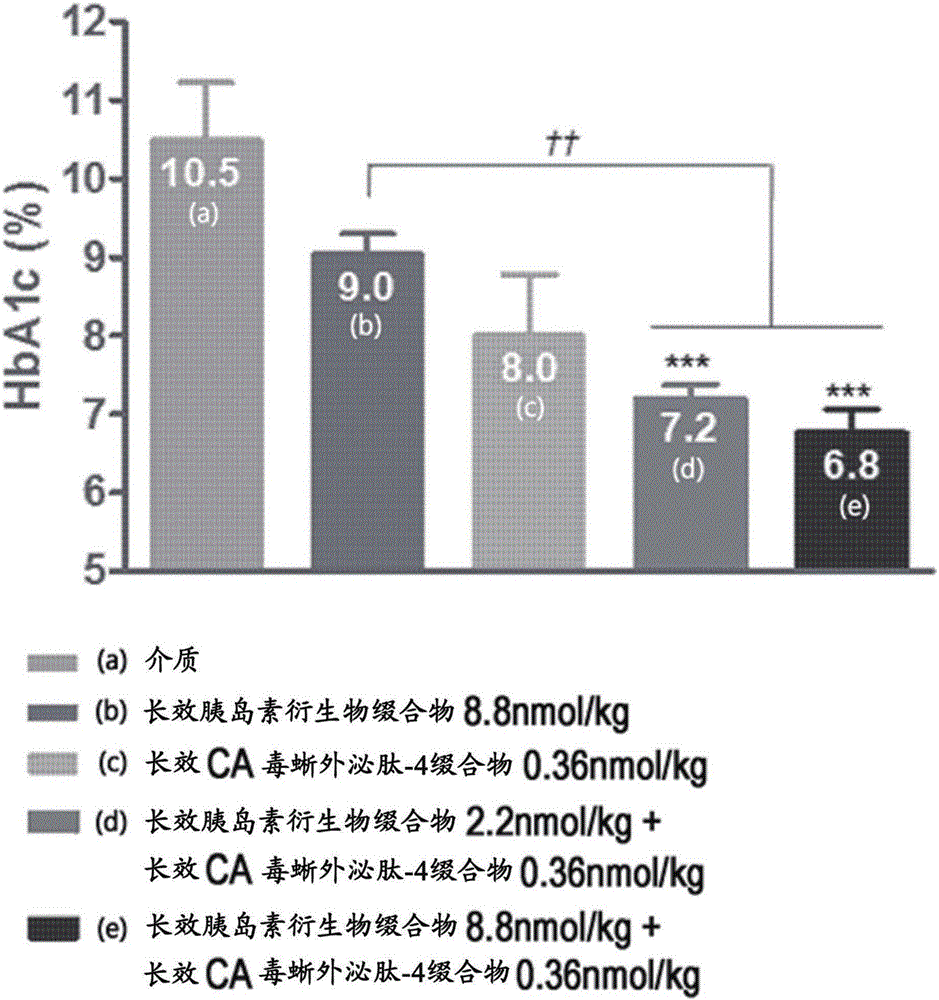
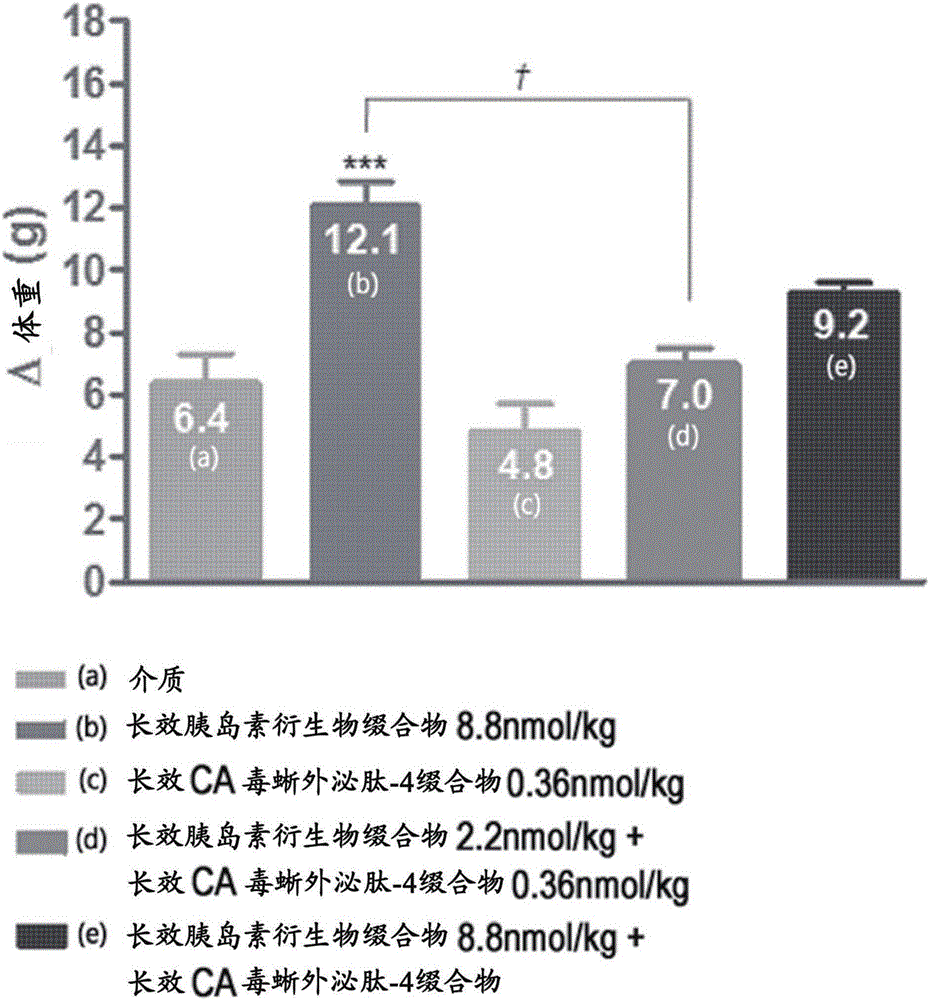
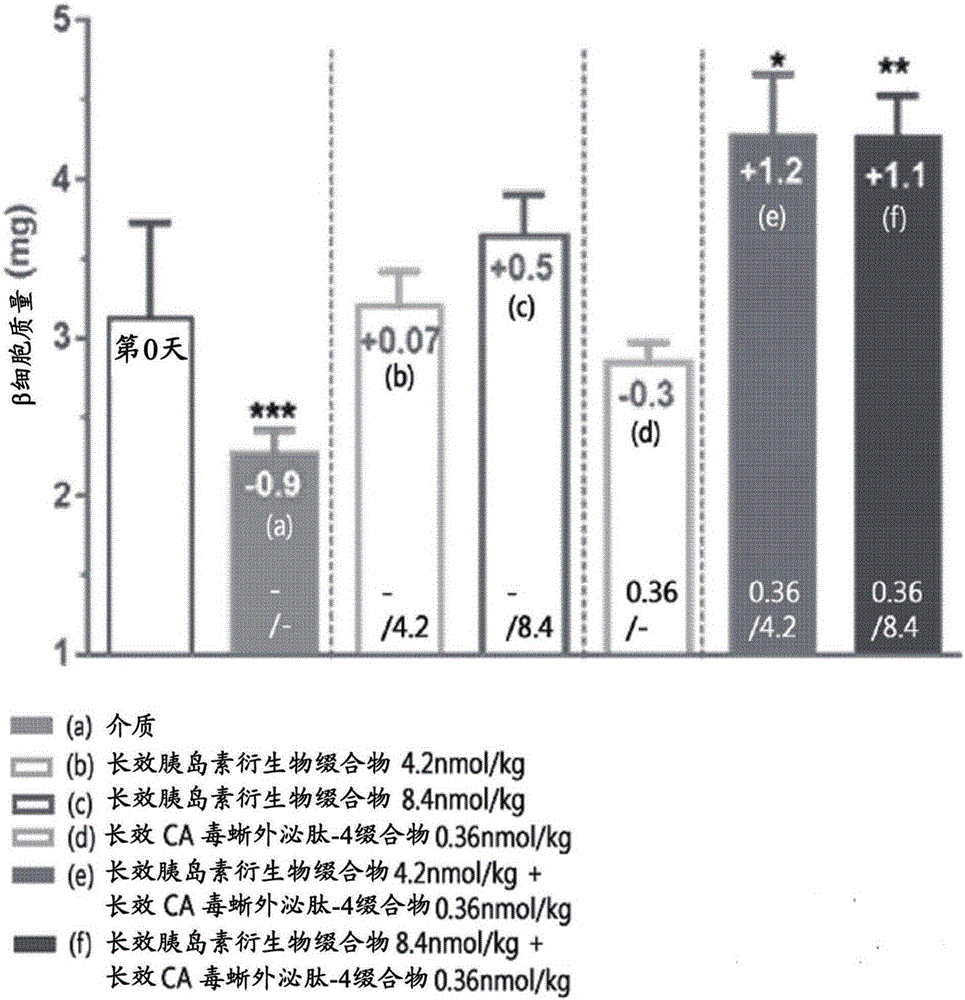
Composition for treating diabetes and containing long-acting insulin analog conjugate and long-acting insulin secretion peptide conjugate
A technology of insulin analogs and insulin-stimulating peptides, which is applied in the field of treating diabetes and reducing the side effects of pancreatic β cells in diabetic patients. It can solve the problems of increasing the half-life of physiologically active polypeptides, so as to improve the duration of efficacy in the body, enhance the duration and stability in the body effect on improving drug adherence
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0133] Embodiment 1: Preparation of single-chain insulin analog expression vector
[0134] To prepare insulin analogs, each with one amino acid change in the A or B chain, forward and reverse oligonucleotides (Table 2) were synthesized using available natural insulin expression vectors as templates, and performed PCR to amplify the individual analog genes.
[0135] The names of each amino acid sequence and each analog modified in the A chain or B chain are listed in Table 1 below. That is, analog 1 substitutes alanine for glycine at position 1 of the A chain, and analog 4 substitutes alanine for glycine at position 8 of the B chain.
[0136] Table 1
[0137] analog modified sequence Analog 1 A 1 G→A
Analog 2 A 2 I→A
Analog 3 A 19 Y→A
Analog 4 B 8 G→A
Analog 5 B 23 G→A
Analog 6 B 24 F→A
Analog 7 B 25 F→A
Analog 8 A 14 Y→E
Analog 9 A 14 Y→N
[0138] Primers use...
Embodiment 2
[0147] Example 2: Expression of Recombinant Insulin Analog Fusion Peptides
[0148] Recombinant insulin analogs were expressed under the control of the T7 promoter. E. coli BL21-DE3 (E. coli B F-dcm ompT hsdS(rB-mB-)gal DE3; Novagen) was transformed with the respective recombinant insulin analog expression vectors. Transformation was performed according to Novagen's recommended procedure. Single colonies transformed with individual recombinant expression vectors were inoculated in 2X Luria Broth (LB) medium containing ampicillin (50 / ml), and cultured at 37°C for 15 hours. Mix each culture solution of the recombinant strain with 2X LB medium containing 30% glycerol at a ratio of 1:1 (v / v), then aliquot 1 ml each into cryopreservation tubes, and store at -140°C . These samples were used as cell stocks for production of recombinant fusion proteins.
[0149] For the expression of recombinant insulin analogs, each 1 vial of cell stock was thawed, then inoculated in 500 ml of ...
Embodiment 3
[0150] Example 3: Recovery and refolding of recombinant insulin analogs
[0151] To convert the recombinant insulin analog expressed in Example 2 into a soluble form, the cells were disrupted and subsequently refolded. 100 g (wet weight) of the cell pellet was resuspended in 1 L of lysis buffer (50 mM Tris-HCl (pH 9.0), 1 mM EDTA (pH 8.0), 0.2M NaCl and 0.5% Triton X-100). Cells were disrupted at 15,000 psi operating pressure using a microfluidizer M-110EH (ACTechnology Corp. Model M1475C). The cell lysates thus disrupted were centrifuged at 7,000 rpm and 4°C for 20 minutes. The supernatant was discarded and the pellet was resuspended in 3 L of wash buffer (0.5% Triton X-100 and 50 mM Tris-HCl (pH 8.0), 0.2 M NaCl, 1 mM EDTA). After centrifugation at 7,000 rpm and 4°C for 20 minutes, the cell pellet was resuspended in distilled water, followed by centrifugation in the same manner. The precipitate thus obtained was resuspended in 400 ml of buffer (1 M glycine, 3.78 g cyste...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More