Preparing method for protein-binding-toxin-imprinted silica gel absorber and adsorption device
A silica gel adsorption and protein technology, applied in chemical instruments and methods, other chemical processes, blood circulation treatment, etc., can solve the problems of unfavorable industrialization, lack of specificity of protein-binding toxins, complex removal process of template molecules, etc., to achieve improved adsorption The effect of high rate, strong permeability and good adsorption performance
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
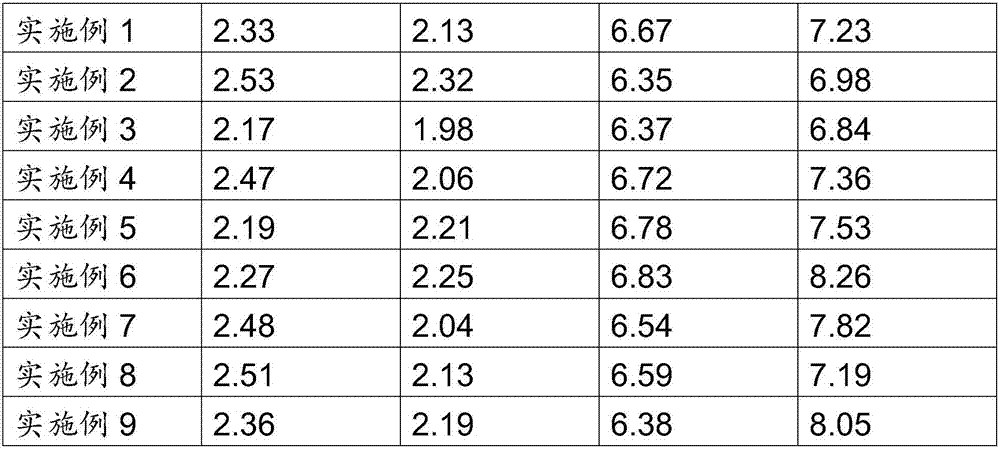
Embodiment 1
[0033] The preparation method of the protein-binding toxin imprinted silica gel adsorbent in this example includes the following steps:
[0034] (1) Mix and disperse 50mL of N-(2-aminoethyl)-3-aminopropyltrimethoxysilane and 150mL tetramethoxysilane at 5°C under nitrogen;
[0035] (2) Weigh the average pore size of 2nm to 3nm, the average particle size of 150μm to 250μm, and the specific surface area of 600m 2 / g to 800m 2 / g, the pore volume is 0.35cm 3 / g to 0.45cm 3 40g of silica gel per g, soaked in 100mL of 6mol / L HCl for 12h, washed with deionized water until neutral, and then dried in a vacuum oven at 40°C for 12h; under normal pressure, soak the obtained silica gel in 50mL of 3mol / L Calcium carbonate aqueous solution, vacuumize, repeat 4 times, filter, dry; And disperse evenly in step (1) gained mixture;
[0036] (3) According to the mass volume ratio of indoxyl sulfate, human serum albumin and deionized water, it is 140.29mg: 30g: 1L to prepare 2mL of protein-bi...
Embodiment 2
[0039] The preparation method of the protein-binding toxin imprinted silica gel adsorbent in this example includes the following steps:
[0040] (1) Mix and disperse 66.67mL of N-(2-aminoethyl)-3-aminopropyltrimethoxysilane and 133.33mL of tetramethoxysilane at -5°C under nitrogen;
[0041] (2) Weigh the average pore size of 2nm to 3nm, the average particle size of 150μm to 250μm, and the specific surface area of 600m 2 / g to 800m 2 / g, the pore volume is 0.35cm 3 / g to 0.45cm 3 45g of silica gel per g, soaked in 100mL of 6mol / L HCl for 12h, washed with deionized water until neutral, and then dried in a vacuum oven at 40°C for 12h; under normal pressure, soak the obtained silica gel in 50mL of 1.5mol / L The calcium carbonate aqueous solution of L, vacuumize, repeat 3 times, filter, dry; And disperse evenly in the mixture gained in step (1);
[0042] (3) According to the mass volume ratio of indoxyl sulfate, human serum albumin and deionized water, it is 95.53mg: 30g: 1L t...
Embodiment 3
[0045] The preparation method of the protein-binding toxin imprinted silica gel adsorbent in this example includes the following steps:
[0046] (1) Mix and disperse 90mL of N-(2-aminoethyl)-3-aminopropyltrimethoxysilane and 110mL tetramethoxysilane at 0°C under nitrogen;
[0047] (2) Weigh the average pore size of 2nm to 3nm, the average particle size of 150μm to 250μm, and the specific surface area of 600m 2 / g to 800m 2 / g, the pore volume is 0.35cm 3 / g to 0.85cm 3 45g of silica gel per g, soaked in 100mL of 6mol / L HCl for 12h, washed with deionized water until neutral, and then dried in a vacuum oven at 40°C for 12h; under normal pressure, soak the obtained silica gel in 50mL of 1.8mol / L The calcium carbonate aqueous solution of L, vacuumize, repeat 4 times, filter, dry; And disperse evenly in the mixture gained in step (1);
[0048] (3) According to the mass volume ratio of indoxyl sulfate, human serum albumin and deionized water, it is 77.94mg: 25g: 1L to prepare ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More