Carbonyl reductase mutant, recombinant expression carrier and application of carbonyl reductase mutant and recombinant expression carrier to production of chiral alcohol
An expression vector and reductase technology, applied in the field of enzyme engineering, can solve the problems of harsh reaction conditions, difficult operation, residual heavy metals, etc., and achieve the effects of easy preparation, environmental friendliness and mild reaction conditions
Active Publication Date: 2019-12-20
ZHEJIANG UNIV +1
View PDF5 Cites 3 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Chemists have found that chiral metal derivatives can be used as catalysts for the asymmetric reduction of carbonyl groups. Although this chemical method has been partially used in industrial production, the method is difficult to operate, the reaction conditions are harsh, and there may be residues in the product Heavy metals, so its application is limited
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
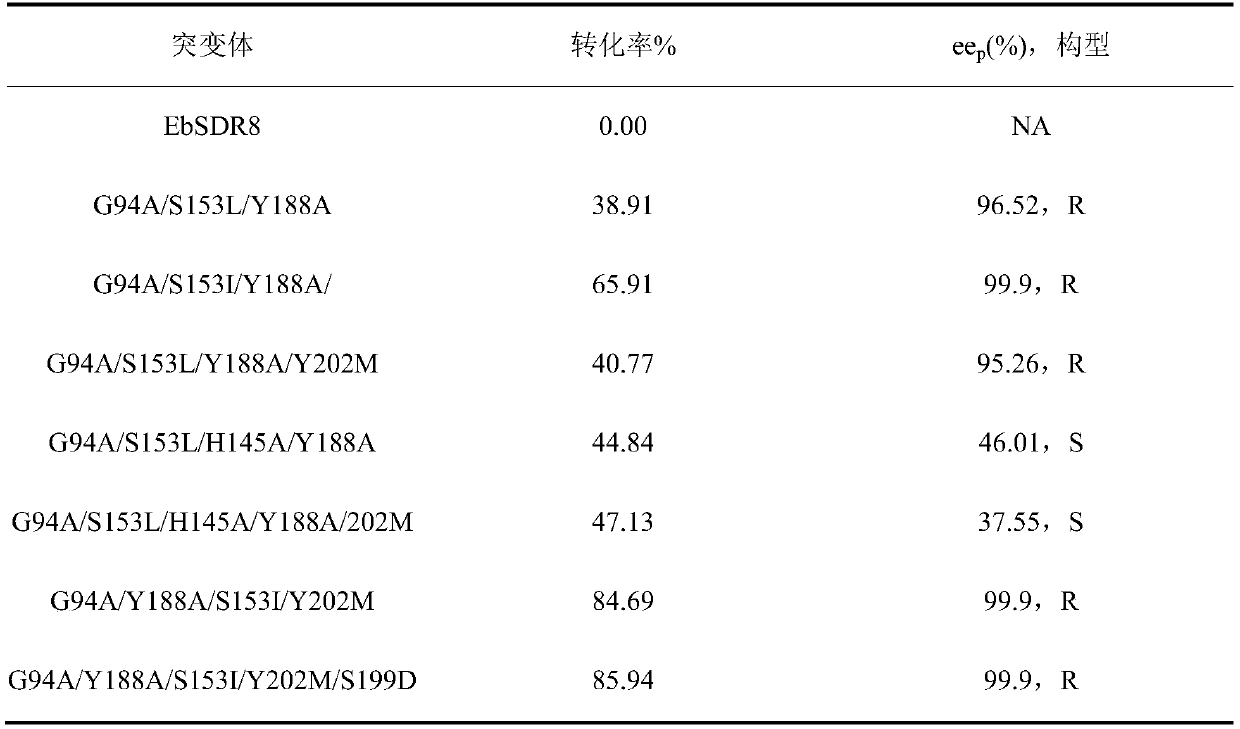
[0027] Example 1, replace the 94th glycine with alanine, the 153rd serine with leucine, and the 188th tyrosine in the amino acid sequence shown in the sequence listing as SEQ ID No.2 Alanine;
Embodiment 2
[0028] Example 2, replace the 94th glycine with alanine, the 188th tyrosine with alanine, and the 153rd serine in the amino acid sequence shown in the sequence listing as SEQ ID No.2 Valine;
Embodiment 3
[0029] Example 3, replacing the glycine at position 94 with alanine, the serine at position 153 with leucine, and the tyrosine at position 188 with Alanine, tyrosine at position 202 is replaced by methionine;
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
| Property | Measurement | Unit |
|---|---|---|
| optical purity | aaaaa | aaaaa |
Login to View More
Abstract
The invention discloses a carbonyl reductase mutant, a recombinant expression carrier and an application of the carbonyl reductase mutant and the recombinant expression carrier to production of chiralalcohol. The mutant is a single-site mutant or multi-site combined mutant containing the following sites based on the amino acid sequence of carbonyl reductase EbSDR8: 94-site glycine, 145-site histidine, 153-site serine, 188-site tyrosine, 199-site serine and 202-site tyrosine. The carbonyl reductase mutant or the recombinant expression carrier containing the mutant can efficiently catalyze asymmetric reduction of high-concentration latent chiral ketone in a reaction system without addition of any coenzymes to generate chiral alcohol high in optical purity (e.e.>99%), and has good industrialization application prospects.
Description
technical field [0001] The invention relates to the technical field of enzyme engineering, in particular to a carbonyl reductase mutant, a recombinant expression vector and its application in the production of chiral alcohol. Background technique [0002] The optically pure chiral alcohol (R)-(3)-chloro-(1)-phenylpropanol (molecular formula is C9H11ClO, molecular mass number is 170.64, CAS-No.: 100306-33) obtained by asymmetric reduction of latent chiral ketone -0) has three active groups (chloro, phenyl, optically pure hydroxyl), and is an important chiral building block for the synthesis of medicine and fine chemicals. Researchers have developed a variety of methods for the synthesis of optically active chiral alcohols, including kinetic resolution and asymmetric synthesis. Among them, using the asymmetric reduction of potential chiral ketones to synthesize optically active chiral alcohols can achieve a theoretical yield of 100%. important method. Chemists have found th...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): C12N9/04C12N15/53C12N15/70C12P7/22
CPCC12N9/0006C12N15/70C12P7/22C12Y101/01184
Inventor 于洪巍邵泽辉
Owner ZHEJIANG UNIV