A pharmaceutical composition and pharmaceutical preparation for promoting blood circulation, removing blood stasis, dredging channels and relieving pain
A pharmaceutical preparation, a technique of promoting blood circulation and removing blood stasis, which is applied in the field of traditional Chinese medicine, can solve the problems of unclear active ingredients, short drug effect duration, and inaccurate drug dosage, so as to achieve clear drug effect material basis, drug effect time, etc. Long-lasting, clear effect of medicinal ingredients
Active Publication Date: 2021-07-23
XIUZHENG PHARMA GROUP HOIDING
View PDF16 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
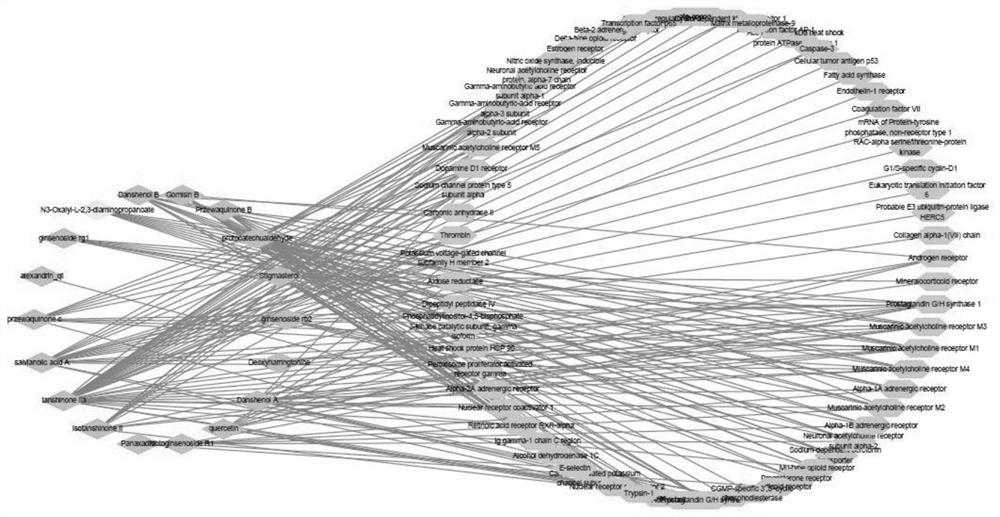
[0003] Danqi Prescription is a classic prescription among medicines for promoting blood circulation, removing blood stasis, dredging channels, and relieving pain. It is composed of Danshen and Panax notoginseng. For formulating prescriptions, the proportions of medicines in each party are different. Although the extracts have been purified and enriched, the extraction and purification methods of the same medicinal materials in various prescriptions are different. There are many chemical components in the extracts and the purity is low. The extraction rate and transfer rate of active ingredients vary greatly, and the extraction rate of active ingredients is also affected by factors such as the source of medicinal materials, batches, processing temperature, and extraction solvents.
The content measurement indicators and limits in the quality control standards of each prescription are also different, and the dosage is not accurate, which leads to uneven clinical efficacy of similar drugs
And only one or a few known components in the extract can be clarified, the active components are not clear, the duration of drug efficacy is short, and the dosage is large, so the impact of unknown components on the efficacy and safety cannot be accurately evaluated
[0004] The existing published patents CN 104415045B, CN 104274518B, and CN 104274520B have complicated components in their compositions. The prescriptions contain 16 known chemical components and borneol in Danshen and Panax notoginseng medicinal materials. The components are complex, the dosage is large, and they cannot fully Explain the correlation between each ingredient in the formula and the efficacy of the medicine
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
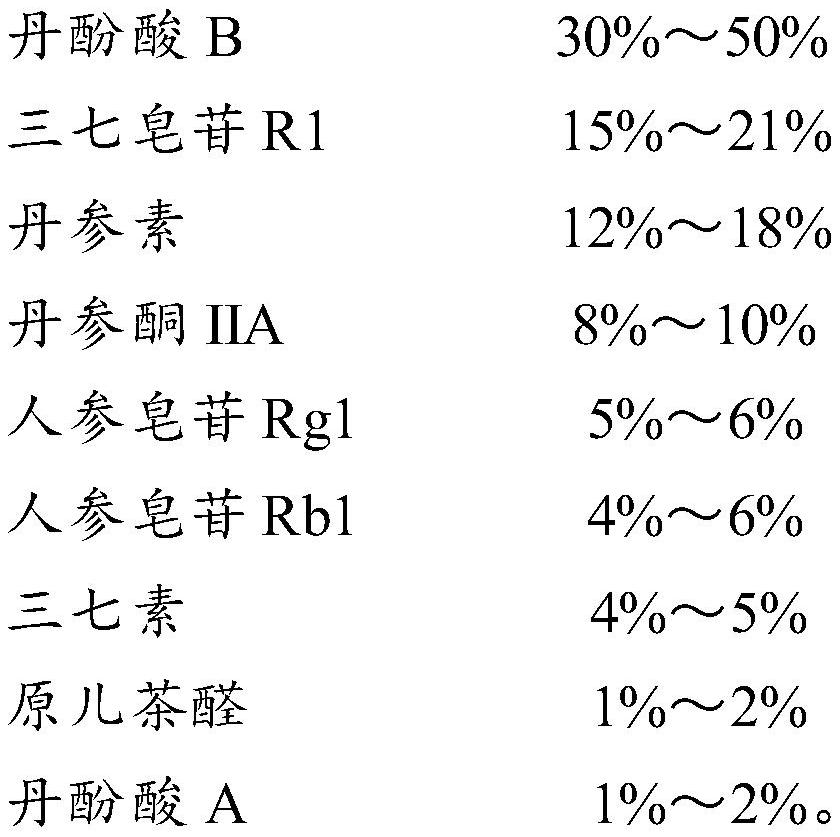
[0051] Take salvianolic acid B 20g, notoginsenoside R1 9g, danshensu 7.5g, tanshinone IIA 4.5g, ginsenoside Rg12.75g, ginsenoside Rb1 2.5g, notoginseng 2.25g, protocatechualdehyde 0.75g, salvianol Acid A 0.75g was mixed uniformly to make 50g.
Embodiment 2
[0053] Take 15g of salvianolic acid B, 10.5g of notoginseng saponin R1, 9g of danshensu, 5g of tanshinone IIA, 3g of ginsenoside Rg1, 3g of ginsenoside Rb1, 2.5g of notoginseng, 1g of protocatechualdehyde, 1g of salvianolic acid A and mix well , made into 50g.
Embodiment 3
[0055] Take salvianolic acid B 25g, notoginsenoside R1 7.5g, danshensu 6g, tanshinone IIA 4g, ginsenoside Rg12.5g, ginsenoside Rb1 2g, notoginseng 2g, protocatechualdehyde 0.5g, salvianolic acid A 0.5g g and mix well to make 50g.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention relates to the technical field of traditional Chinese medicine, in particular to a pharmaceutical composition and a pharmaceutical preparation for promoting blood circulation, removing blood stasis, dredging pulses and relieving pain. By weight percentage, by weight percentage, by salvianolic acid B 20%~60%, notoginseng saponin R1 10%~25%, danshensu 10%~20%, tanshinone IIA 5%~12%, ginsenoside Rg1 1%-10%, ginsenoside Rb1 1%-10%, notoginseng 1%-10%, protocatechualdehyde 0.5%-2.5%, salvianolic acid A 0.5%-2.5%. The invention formulates the prescription according to the scientific ratio of the medicine with high purity and clear pharmacological action, safe dose and toxic and side effects, and provides a medicinal drug with reasonable prescription, clear medicinal effect components, few impurities, fast onset of effect, A medicinal composition with the functions of promoting blood circulation and removing blood stasis, clearing the pulse and relieving pain, taking a small dose, and having a clear medicinal material basis, precise medication and precise treatment.
Description
technical field [0001] The invention relates to the technical field of traditional Chinese medicine, in particular to a pharmaceutical composition and pharmaceutical preparation for promoting blood circulation, removing blood stasis, dredging channels and relieving pain. Background technique [0002] Blood stasis syndrome is a common clinical disease, and its onset is caused by slow blood flow and blockage of veins due to various stasis-causing factors. According to different pathogenic factors, it can be divided into cold coagulation blood stasis, qi stagnation blood stasis, qi deficiency blood stasis and other types. The lesions can affect multiple systems such as the heart, brain, lung, kidney, liver, and reproduction. In mild cases, it can affect the daily life of patients. life, and seriously endanger the patient's life. Therefore, research on blood stasis syndrome and drugs for promoting blood circulation and removing stasis is one of the most urgent contents in the f...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Patents(China)
IPC IPC(8): A61K31/704A61K31/343A61K31/192A61K31/58A61K31/198A61K31/11A61K31/216A61P29/00
CPCA61K31/11A61K31/192A61K31/198A61K31/216A61K31/343A61K31/58A61K31/704A61P29/00A61K2300/00
Inventor 吴萍曲笑锋徐建赵伯友于艳辉高嵩
Owner XIUZHENG PHARMA GROUP HOIDING