Sustained-release drug delivery system
A drug and pharmacy technology, applied in the direction of drug delivery, pharmaceutical formulation, aerosol delivery, etc., can solve the problems of pain and irritation at the injection site, oil gel preparations that have not been seen, preparation application restrictions, etc., to achieve good biophase Compatibility, good drug safety and tolerability, and ease of administration
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0102] The dissolving situation investigation of embodiment 1 different types of saturated phospholipids in castor oil and each organic solvent
[0103]Take 1g of castor oil and organic solvent respectively in an EP tube, add 0.1g of saturated phospholipid (about 100mg / g, 10% w / w), ultrasonicate at 50°C, and observe the dissolution. The results are shown in Table 1-1.
[0104] Table 1-1 Dissolution investigation
[0105]
[0106] Among them, EtOH represents absolute ethanol; BB represents benzyl benzoate; BA represents benzyl alcohol; NMP represents N-methylpyrrolidone; DMSO represents dimethyl sulfoxide.
[0107] According to the dissolution investigation, saturated phospholipids (HSPC, DPPC, DMPC, DSPC) have good solubility in alcohol solvents (ethanol, benzyl alcohol, propylene glycol, etc.), while phosphatidylglycerol (DPPG), phosphatidylethanolamine (DPPE) , Phosphatidic acid (DPPA) can not be dissolved in the above solvents.
Embodiment 2
[0108] Embodiment 2 composition ratio research
[0109] (1) Morphological test of the composition at room temperature
[0110] Compositions were prepared according to each ingredient and ingredient ratio shown in Table 2-1 below. Mix the solvent phospholipid, oil and solvent, stir while heating until a transparent and uniform solution is formed, cool to room temperature, and examine the physical form. At the same time, the room temperature morphology of the unsaturated phospholipid composition was used as a comparative study.
[0111] Table 2-1 The room temperature form of the composition under different proportions
[0112]
[0113] In this example, the preparation form of the composition containing 0.5-30% (w / w) saturated phospholipids (HSPC, DPPC, DSPC and DMPC) and 0-50% (w / w) solvent at room temperature is investigated, and the results are shown in Table 2 As shown in -1, the room temperature form of the composition of the present invention is related to the amount ...
Embodiment 3
[0122] (1) Effects of different types of phospholipids on the phase transition of the composition
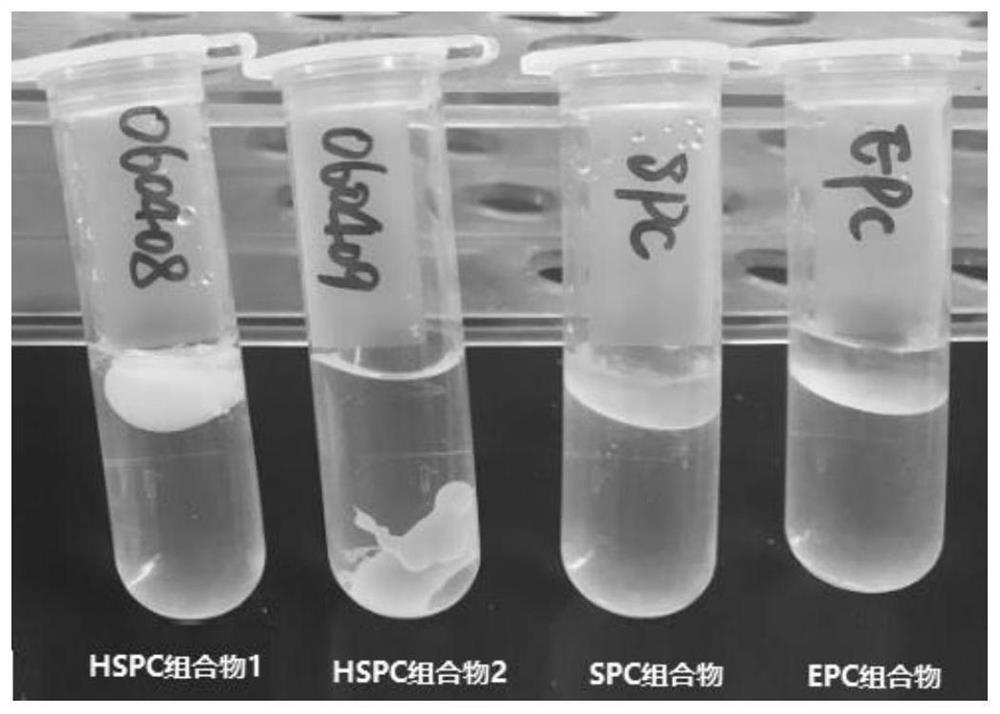
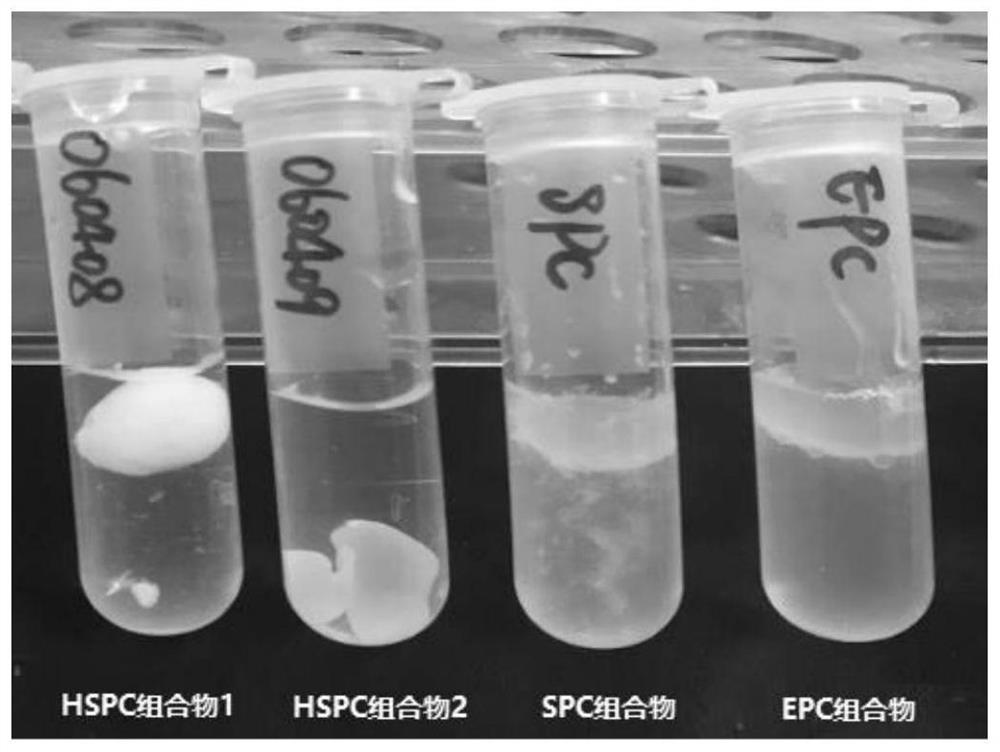
[0123] According to Table 3-1, pharmaceutical compositions containing saturated phospholipids (composition 16), phospholipid-free (pure castor oil group) and unsaturated phospholipids were prepared respectively. Dissolve meloxicam in N-methylpyrrolidone to prepare a concentrated solution of 50 mg / g, add ropivacaine, soybean lecithin or dipalmitoyl phosphatidylcholine to liquid oil, solvent and meloxime at 50°C In the Kang solution, stir while heating until a transparent and uniform solution is formed, and cool to room temperature. Slowly inject the composition into water with a syringe, and observe the morphological changes of different compositions in water. see results Picture 1-1 .
[0124] Table 3-1 Different Compositions
[0125]
[0126] Above-mentioned experiment has investigated the morphological change in water containing saturated phospholipid (composition 16), ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Viscosity | aaaaa | aaaaa |
| Viscosity | aaaaa | aaaaa |
| Viscosity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More