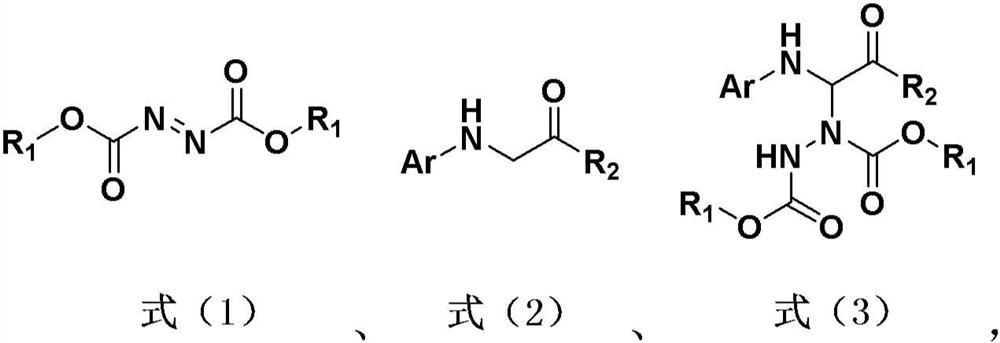
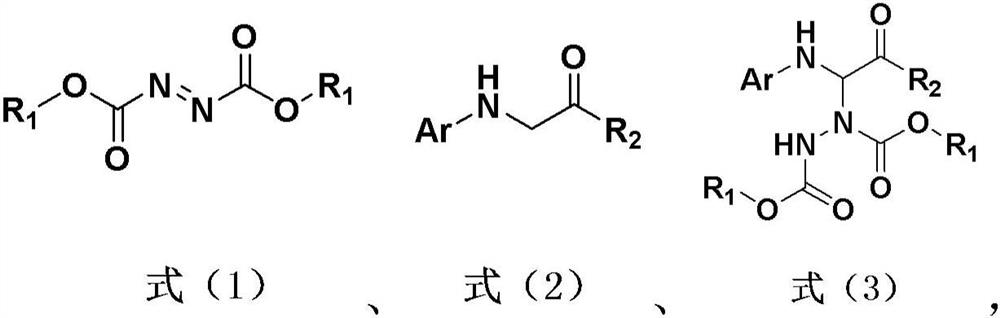
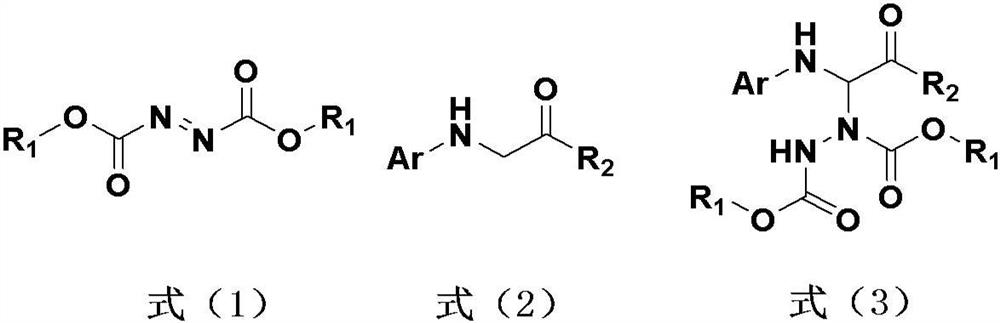
Hydrazine modification method of visible light induced glycine derivative
A modification method, glycine technology, which is applied in the field of hydrazine modification of glycine derivatives induced by visible light, can solve the problems of narrow substrate range and limited industrial application, and achieve high selectivity, no catalyst and additives, and high atom utilization. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0039] Reaction formula:
[0040]
[0041] The specific method is as follows: Add azobis-tert-butyl ester (0.3 mmol) and N-phenylglycine to a 10 mL Schlenk reaction tube (Beijing Xinweier Glass Instrument Co., Ltd., F891410 reaction tube, capacity 10 mL, ground 14 / 20) Ethyl ester (0.2 mmol). The air in the tube was completely replaced with argon three times, and then 1 mL of acetonitrile was added under an argon atmosphere. The reaction system was continuously stirred at room temperature for 12 hours under the irradiation of a 40W purple LED (427nm) lamp (IKA magnetic stirrer, RCT basic type, stirring speed 500 rpm). After the reaction is complete, use H 2 The reaction was quenched with O, and the reaction solution was extracted with ethyl acetate (3*10 mL), and then the combined organic phase was concentrated by rotary evaporation (BUCHI Co., Ltd., BUCHI rotary evaporator R-3). The concentrated residue is passed through a chromatographic column (Beijing Xinweier Glass I...
Embodiment 2
[0047] Reaction formula:
[0048]
[0049] The method is the same as in Example 1, the difference lies in the type of glycine derivative shown in the above formula, and the yield is shown in Table 1.
[0050] Gained product is carried out nuclear magnetic resonance analysis, the result is as follows:
[0051] 1 H NMR (400MHz, CDCl 3 )δ6.72-6.66 (m, 4H), 6.30-5.79 (m, 2H), 4.18 (d, J=7.0Hz, 2H), 3.67 (s, 3H), 1.50-1.32 (m, 18H), 1.26 (t, J=7.1Hz, 3H).
[0052] 13 C NMR (101MHz, CDCl 3 )δ167.8, 154.6, 153.2, 137.8, 115.2, 114.9, 82.1, 81.2, 65.9, 62.2, 55.7, 28.1, 27.9, 14.0.
[0053] HRMS (ESI) calcd for C 21 h 34 N 3 o 7 + [M+H] + :440.2391, found: 440.2394.
Embodiment 3
[0055] Reaction formula:
[0056]
[0057] The method is the same as in Example 1, the difference lies in the type of glycine derivative shown in the above formula, and the yield is shown in Table 1.
[0058] Gained product is carried out nuclear magnetic resonance analysis, the result is as follows:
[0059] 1 H NMR (400MHz, CDCl 3 )δ6.90(t, J=8.6Hz, 2H), 6.71(s, 2H), 6.14-5.93(m, 2H), 4.25-4.21(m, 2H), 1.60-1.30(m, 21H).
[0060] 13 C NMR (101MHz, CDCl 3 )δ167.8, 156.7(d, J=236.9Hz), 140.3, 123.4(d, J=8.7Hz), 115.8(d, J=22.4Hz), 114.9, 82.3, 81.4, 66.0, 62.4, 28.1, 27.9 , 14.0.
[0061] HRMS (ESI) calcd for C 20 h 31 FN 3 o 6 + [M+H] + : 428.2191, found: 428.2195.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com