Synthesis and purification method of high-content chloroformic acid-2-ethyl hexyl ester
A technology of ethylhexyl ester and purification method, applied in the purification/separation of carbonate/haloformate, preparation of phosgene or haloformate, organic chemistry, etc., can solve the problems of low purity and content, Achieve high product content, reduce post-treatment energy consumption, and inhibit side reactions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
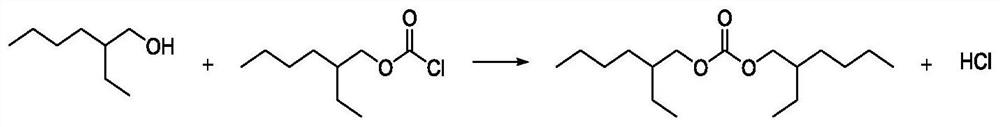
[0040] Put 20.0 mol of iso-octanol into the synthesis kettle, the ratio of phosgene:iso-octanol is 1:1.5, the reaction time is 4 hours, and the reaction temperature is 15°C. After the reaction in the low temperature section, the analytical content of the synthetic solution was 97.6% of 2-ethylhexyl chloroformate, 0.27% of diisooctyl carbonate, 1.16% of isooctyl alcohol, and 0.97% of chloroisooctane. Raise the temperature of the synthesis liquid in the low-temperature section to 40°C, and feed in phosgene. The molar ratio of phosgene to iso-octanol is 0.2:1, and the reaction time is 2 hours. After the reaction in the high temperature section, the analytical content of the synthetic solution was 98.2% of 2-ethylhexyl chloroformate, 0.29% of diisooctyl carbonate, 0.01% of isooctyl alcohol, and 1.5% of chloroisooctane. Light, acidity less than 0.1%.
[0041] The synthetic liquid of 2-ethylhexyl chloroformate extracted from the synthetic kettle is sent to the mixing kettle, and an...
Embodiment 2
[0043] Put 20.0 mol of isooctyl alcohol into the synthesis kettle, the ratio of phosgene:isooctanol is 1:1.3, the reaction time is 4 hours, and the reaction temperature is 15°C. After the reaction in the low temperature section, the analytical content of the synthetic liquid was 96.51% of 2-ethylhexyl chloroformate, 0.30% of di-isooctyl carbonate, 2.3% of isooctyl alcohol, and 0.89% of chloroisooctane. Raise the temperature of the synthesis liquid in the low-temperature section to 40°C, and feed in phosgene. The molar ratio of phosgene to iso-octanol is 0.2:1, and the reaction time is 2 hours. After the reaction in the high temperature section, the analytical content of the synthetic solution was 97.2% of 2-ethylhexyl chloroformate, 0.52% of diisooctyl carbonate, 0.005% of isooctyl alcohol, and 2.27% of chloroisooctane. Light, acidity less than 0.1%.
[0044] Send the synthesis liquid of 2-ethylhexyl chloroformate into the mixing tank, add an entrainer, the mass ratio of DMF:...
Embodiment 3
[0046] Put 20.0 mol of iso-octanol into the synthesis kettle, the ratio of phosgene:iso-octanol is 1:1.5, the reaction time is 6 hours, and the reaction temperature is 15°C. After the reaction in the low temperature section, the analytical content of the synthetic solution was 97.8% of 2-ethylhexyl chloroformate, 0.32% of diisooctyl carbonate, 0.93% of isooctyl alcohol, and 0.95% of chloroisooctane. Raise the temperature of the synthesis solution in the low-temperature section to 50°C, and feed in phosgene. The molar ratio of phosgene to iso-octanol is 0.2:1, and the reaction time is 2 hours. After the reaction in the high temperature section, the analytical content of the synthetic solution was 98.4% of 2-ethylhexyl chloroformate, 0.69% of diisooctyl carbonate, 0.01% of isooctyl alcohol, and 0.90% of chloroisooctane. Light, acidity less than 0.1%.
[0047] Send the synthesis liquid of 2-ethylhexyl chloroformate into the mixing tank, add an entrainer, the mass ratio of DMF:sy...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More