Carbon quantum dot modified dual-network hydrogel and preparation method thereof
A carbon quantum dot and double network technology, applied in the field of carbon quantum dot modified double network hydrogel and its preparation, can solve problems such as human health and environmental hazards, achieve control of water loss, promote cell proliferation, and accelerate wound healing Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0035] The preparation method of the carbon quantum dot modified double network hydrogel of the present embodiment comprises:
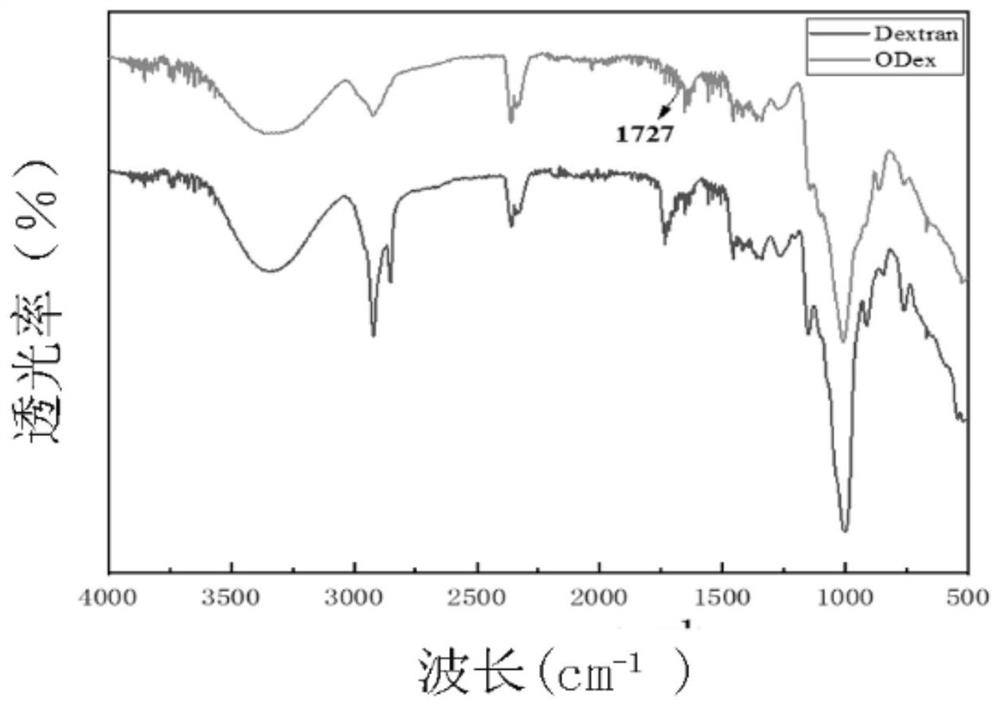
[0036] Step 1. Mix the oxidized dextran aqueous solution and the recombinant collagen aqueous solution to obtain the oxidized dextran-recombinant collagen mixed solution; the mass percentage of the oxidized dextran aqueous solution is 6%, and the oxidized glucan The number average molecular weight of the polysaccharide is 40000 Da, the mass percentage of the recombinant collagen in the recombinant collagen aqueous solution is 6%, and the volume ratio of the oxidized dextran aqueous solution and the recombinant collagen aqueous solution is 1:1;
[0037] Step 2: Dissolve kanamycin in the reaction kettle, treat at 160°C for 2 hours, then centrifuge at 15,000 rpm for 30 minutes, dialyze the centrifugate for 3 days with a dialysis bag with a cut-off of 1,000 Da, and filter it with a 0.22 μm filter membrane After lyophilization for 3 days, kanamycin carbon ...
Embodiment 2
[0042] The preparation method of the carbon quantum dot modified double network hydrogel of the present embodiment comprises:
[0043] Step 1. Mix the oxidized dextran aqueous solution and the recombinant collagen aqueous solution to obtain the oxidized dextran-recombined collagen mixed solution; the mass percentage of the oxidized dextran aqueous solution is 10%, and the oxidized glucan The number average molecular weight of the polysaccharide is 40000Da, the mass percentage of the recombinant collagen in the recombinant collagen aqueous solution is 8%, and the volume ratio of the oxidized dextran aqueous solution and the recombinant collagen aqueous solution is 1:1.5;
[0044] Step 2: Dissolve kanamycin in the reaction kettle, treat at 165°C for 2 hours, then centrifuge at 15,000 rpm for 30 minutes, dialyze the centrifugate for 3 days with a dialysis bag with a cut-off of 1,000 Da, and filter it with a 0.22 μm filter membrane Freeze-dry for 3 days to obtain kanamycin carbon ...
Embodiment 3
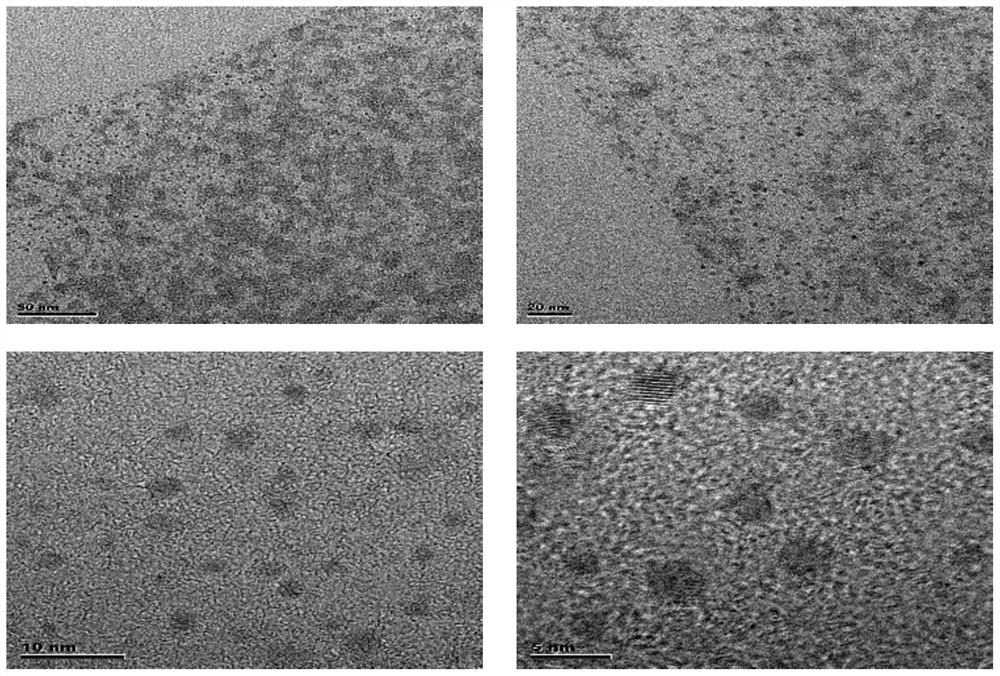
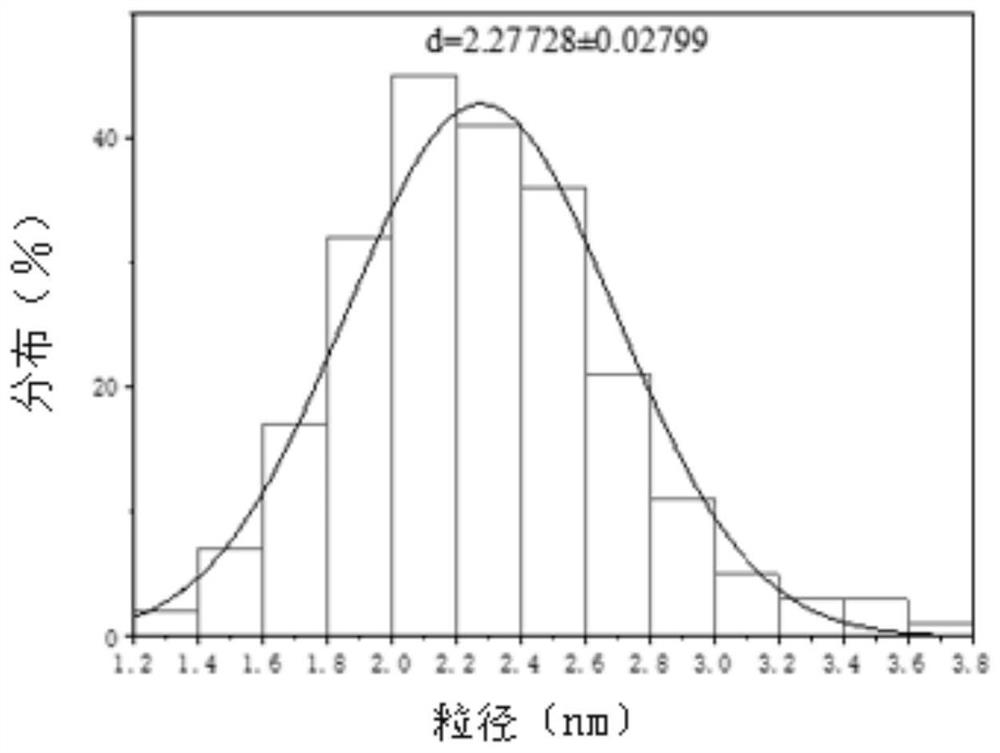
[0049] The double-network hydrogel modified by carbon quantum dots in this embodiment is prepared by introducing kanamycin carbon quantum dots into the double-network hydrogel system by chemical cross-linking; the double-network hydrogel modified by carbon quantum dots The pore size of the gel is 10 μm to 20 μm, and the size of the kanamycin carbon quantum dot is 1 nm to 5 nm.
[0050] The preparation method of the carbon quantum dot modified double network hydrogel of the present embodiment comprises:
[0051] Step 1. Mix the oxidized dextran aqueous solution and the recombinant collagen aqueous solution to obtain the oxidized dextran-recombined collagen mixed solution; the mass percentage of the oxidized dextran aqueous solution is 10%, and the oxidized glucan The number average molecular weight of the polysaccharide is 40000 Da, the mass percentage of the recombinant collagen in the recombinant collagen aqueous solution is 10%, and the volume ratio of the oxidized dextran a...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Aperture | aaaaa | aaaaa |
| Size | aaaaa | aaaaa |
| Number average molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More