Method for preparing oxazolidine
A technology of oxazolidine and raw materials, which is applied in the field of preparation of oxazolidine, can solve the problems of system gel, low stability, disadvantages, etc., and achieve the effect of expanding the scope of use
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
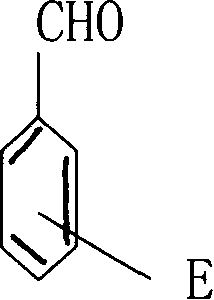
[0022] Embodiment 1: a kind of preparation method of oxazolidine, comprising (1) raw material selection: raw material comprises: the carbonyl compound with benzene ring, and its structural formula is:
[0023] Wherein, E represents phenyl, and diethanolamine, and the molar ratio of the two is 1: 1; (2) mixing process: adding diethanolamine to the reactor, then, the above-mentioned carbonyl compound with a benzene ring is added according to the above mole The equivalence ratio is slowly added to the reactor to form a mixed solution with diethanolamine. During the addition process, the temperature of the mixed solution in the reactor is always kept in the range of 30°C to 45°C; (3) Reaction process: when the temperature is 50°C Under the condition of ℃, the mixed solution was reacted, and the reaction time was: 1 hour; (4) suction filtration process: the water content in the reacted liquid in the reactor was extracted with a vacuum suction filtration device, and then the reacto...
Embodiment 2
[0024] Embodiment 2: a kind of preparation method of oxazolidine, including (1) raw material selection: raw material comprises: the carbonyl compound with benzene ring, and its structural formula is:
[0025] Wherein, E represents benzyl, and N,N'-dihydroxyethylethylenediamine, the molar ratio of the two is 1:2; (2) mixing process: N,N'-dihydroxyethylethylenediamine The amine is added to the reactor, then, the above-mentioned carbonyl compound with a benzene ring is slowly added to the reactor according to the above-mentioned molar equivalent ratio to form a mixed solution with N,N'-dihydroxyethylethylenediamine. During the adding process, Always keep the temperature of the mixed solution in the reactor in the range of 30°C-45°C; (3) reaction process: under the condition that the temperature is 80°C, the mixed solution is reacted, and the reaction time is: 2 hours; ( 4) Suction filtration process: use a vacuum suction filtration device to extract the moisture in the reaction...
Embodiment 3
[0026] Embodiment 3: a kind of preparation method of oxazolidine, including (1) raw material selection: raw material comprises: the carbonyl compound with benzene ring, and its structural formula is:
[0027] Wherein, E represents phenoxy, and 2-amino-2-hydroxymethyl-1,3-propanediol, and the molar ratio of the two is 1:2; (2) mixing process: 2-amino-2-hydroxymethyl base-1,3-propanediol is added to the reactor, then, the above-mentioned carbonyl compound with a benzene ring is slowly added to the reactor according to the above molar equivalent ratio and 2-amino-2-hydroxymethyl-1,3-propanediol A mixed solution is formed, and during the addition process, the temperature of the mixed solution in the reactor is always kept in the range of 30°C-45°C; (3) Reaction process: under the condition that the temperature is 65°C, the mixed solution is reacted , the reaction time is: 1.5 hours; (4) suction filtration process: use the vacuum suction filtration device to extract the moisture ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com