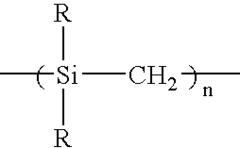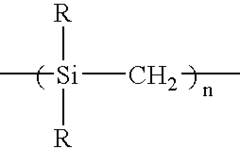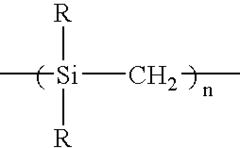Silicon carbide fiber having boron nitride layer in fiber surface and process for the production thereof
a technology of boron nitride and fiber surface, which is applied in the field of silicon carbide fiber, can solve the problems of high cost of process, high cost of raw material gas, and very brittle breakdown behavior of materials,
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
referential example 1
2.5 liters of anhydrous toluene and 400 g of metallic sodium were placed in a three-necked flask having a volume of 5 liters, the mixture was heated to the boiling point of toluene under a flow of nitrogen gas, and 1 liter of dimethyldichlorosilane was dropwise added over 1 hour. After the completion of the addition, the mixture was refluxed under heat for 10 hours to obtain a precipitate. The precipitate was recovered by filtration, and washed with methanol and then with water to give 420 g of a white powder polydimethylsilane.
250 g of the polydimethylsilane was placed in a three-necked flask equipped with a water-cooling refluxing device, and allowed to react under heat at 420° C. for 30 hours under a nitrogen flow, to obtain a polycarbosilane having a number average molecular weight of 1,200.
example 1
100 g of toluene and 100 g of tributyl borate were added to 100 g of polycarbosilane synthesized according to Referential Example 1, the mixture was preliminarily heated at 100° C. for 1 hour, then the mixture was slowly temperature-increased up to 150° C. to distill the toluene off, the resultant mixture was allowed to react at the above temperature for 5 hours, and then the reaction mixture was further temperature-increased up to 250° C. and allowed to react at this temperature for 5 hours, to obtain a modified polycarbosilane. 10 g of tributyl borate was added to the modified polycarbosilane for the purpose of intentionally making a low-molecular weight organic boron compound coexist, to obtain a mixture of the modified polycarbosilane and the low-molecular weight organic boron compound.
The mixture of the modified polycarbosilane and the low-molecular weight organic boron compound was dissolved in toluene, the resultant solution was placed in a spinning machine made of glass, ...
example 2
10 g of aluminum acetylacetonate was added to a toluene solution in which 100 g of polycarbosilane synthesized according to Referential Example 1 was dissolved, and the mixture was allowed to react in a crosslinking reaction at 320° C. under a nitrogen gas flow, to obtain a polyaluminocarbosilane having a number average molecular weight of 2,000.
100 g of toluene and 100 g of tributyl borate were added to 100 g of the obtained polyaluminocarbosilane, the mixture was preliminarily heated at 100° C. for 1 hour, then the mixture was slowly temperature-increased up to 150° C. to distill the toluene off, the resultant mixture was allowed to react at the above temperature for 5 hours, and then the reaction mixture was further temperature-increased up to 250° C. and allowed to react at this temperature for 5 hours, to obtain a modified polycarbosilane. 10 g of tributyl borate was added to the modified polycarbosilane for the purpose of intentionally making a low-molecular weight organic ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Length | aaaaa | aaaaa |
| Percent by mass | aaaaa | aaaaa |
| Percent by mass | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More