Radiolucent spinal fusion cage
a fusion cage and radiolucent technology, applied in the field of bone graft improvement, can solve the problems of potential graft necrosis, poor radiolucency characteristics of traditional titanium-based implant devices, and difficulty in post-operative monitoring and evaluation of the fusion process, and achieve enhanced bone ingrowth and fusion, high porosity, and high strength loading
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
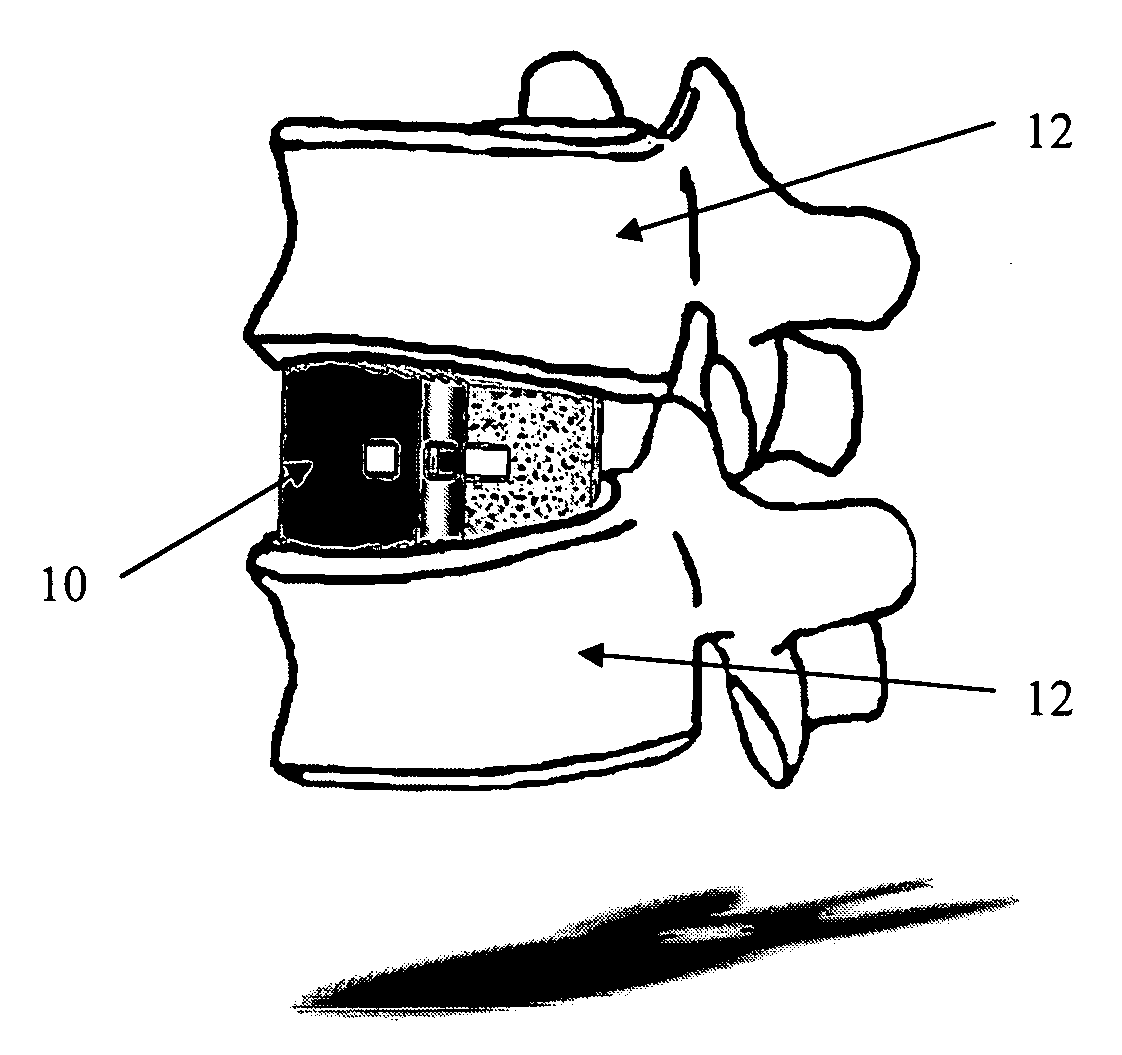
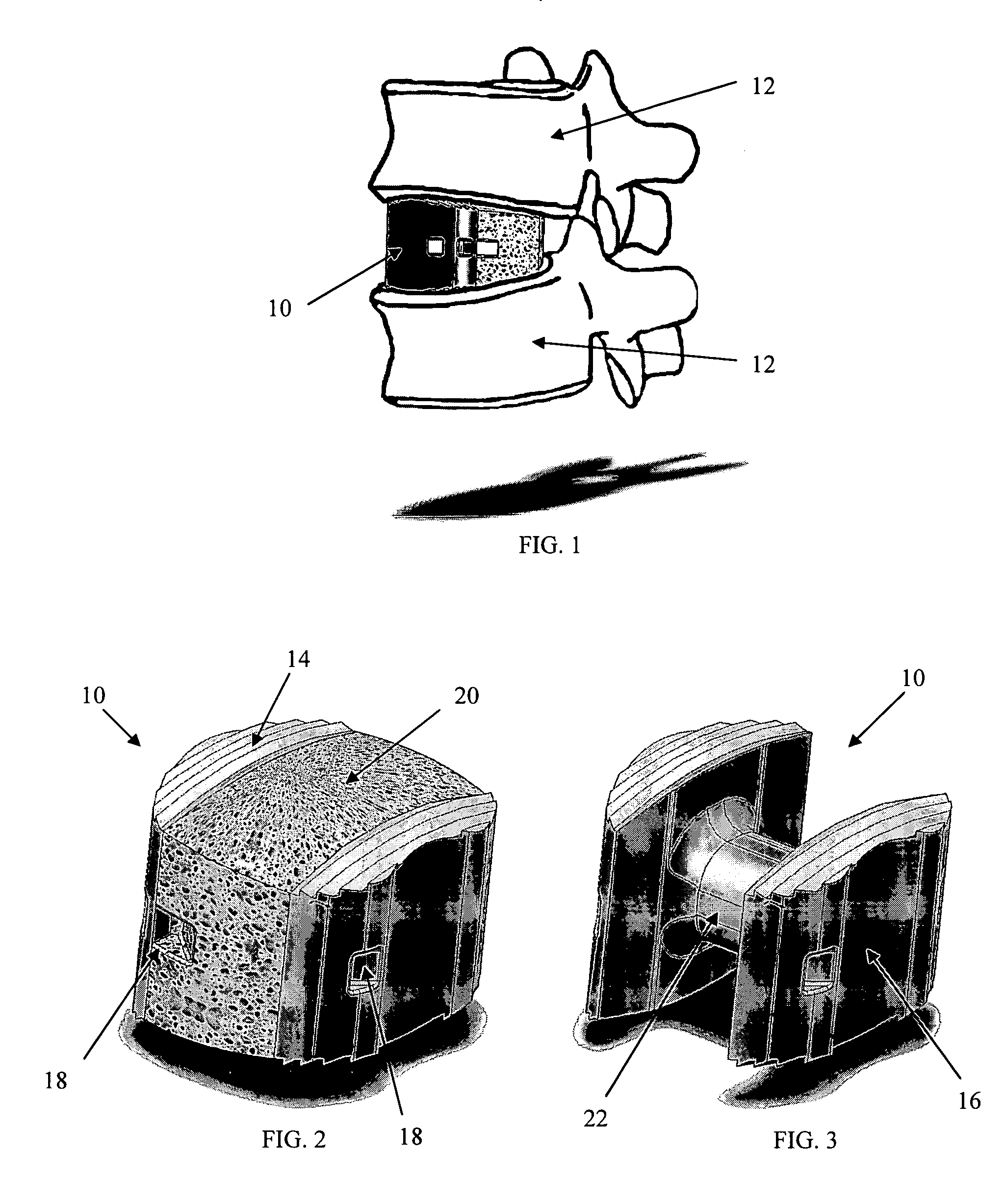
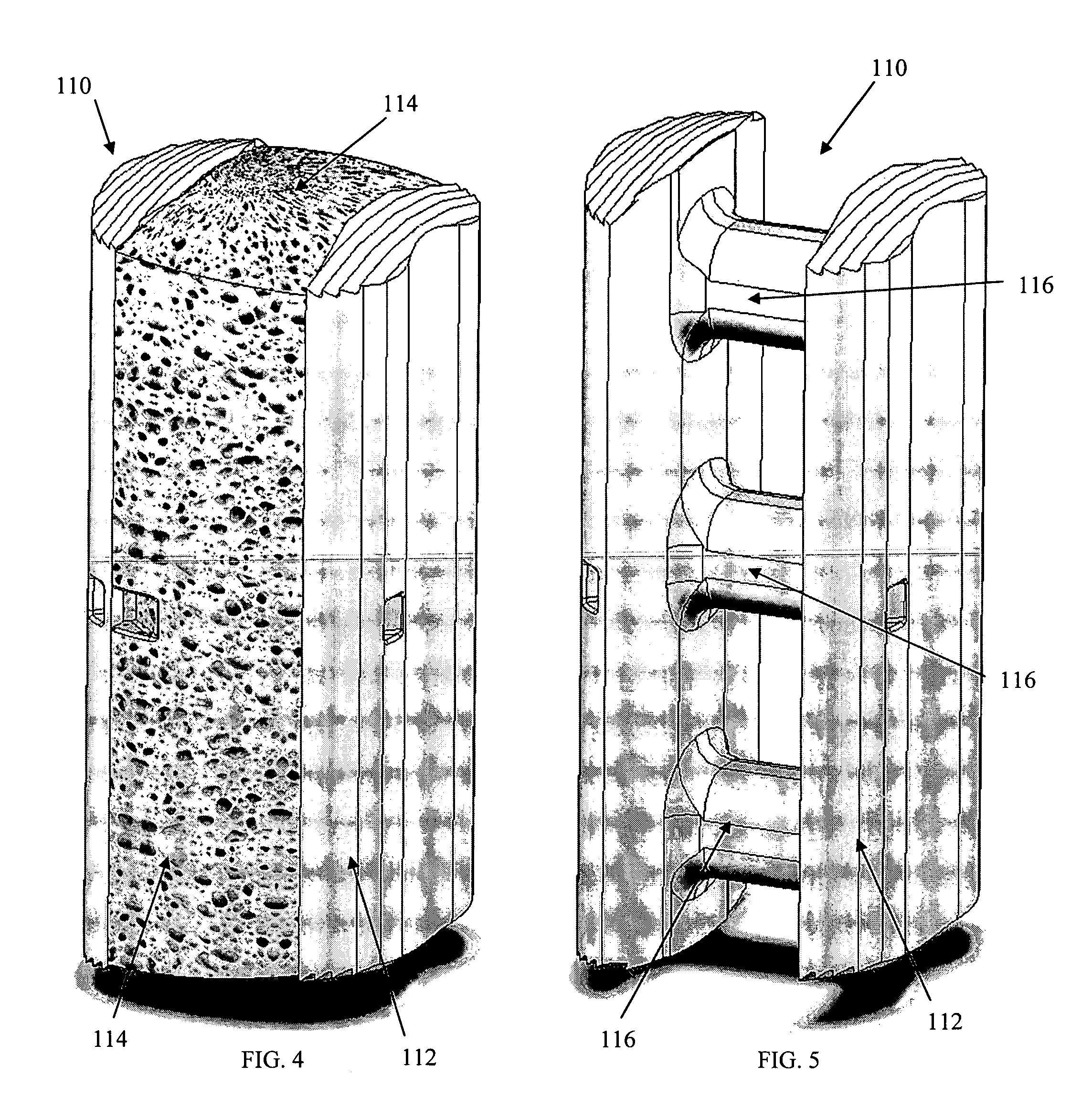
[0033] As shown in the exemplary drawings, a radiolucent bone graft such as a spinal fusion cage referred to generally in FIGS. 1-3 by the reference numeral 10 is provided for seated implantation between a pair of adjacent patient bones such as spinal vertebrae 12 (FIG. 1) to maintain the vertebrae in spaced relation while promoting interbody bone ingrowth and fusion. In general, the improved bone graft 10 comprises a bio-compatible substrate having a porous construction to define an open lattice conducive to interbody bone ingrowth and fusion, while providing a strong mechanical load bearing structure analogous to the load bearing properties of cortical and cancellous bone. This open-celled substrate is coated internally and externally with a bio-active surface coating selected for relatively strong osteoconductive and osteoinductive properties, whereby the coated substrate provides a scaffold conducive to cell attachment and proliferation to promote interbody bone ingrowth and fus...
PUM
| Property | Measurement | Unit |
|---|---|---|
| pore size | aaaaa | aaaaa |
| pore sizes | aaaaa | aaaaa |
| pore sizes | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More