Antigen arrays for treatment of allergic eosinophilic diseases
an array and antibody technology, applied in the field of molecular biology, virology, immunology and medicine, can solve the problems of asthmatic patients refractory to the use of corticosteroids, high cost of monoclonal antibodies, and patient compliance, and achieve the effect of inhibiting eosinophilia and inducing high titers
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Construction and Expression of Mutant Qβ Coat Proteins, and Purification of Mutant Qβ Coat Protein VLPs or Capsids
Plasmid Construction and Cloning of Mutant Coat Proteins
Construction of pQβ-240:
[0272] The plasmid pQβ10 (Kozlovska, T M, et al., Gene 137:133-137) was used as an initial plasmid for the construction of pQβ-240. The mutation Lys13→Arg was created by inverse PCR. The inverse primers were designed in inverted tail-to-tail directions:
(SEQ ID NO:356)5′-GGTAACATCGGTCGAGATGGAAAACAAACTCTGGTCC-3′and(SEQ ID NO:357)5′-GGACCAGAGTTTGTTTTCCATCTCGACCGATGTTACC-3′.
[0273] The products of the first PCR were used as templates for the second PCR reaction, in which an upstream primer
5′-AGCTCGCCCGGGGATCCTCTAG-3′(SEQ ID NO:358)
[0274] and a downstream primer
(SEQ ID NO:359)5′-CGATGCATTTCATCCTTAGTTATCAATACGCTGGGTTCAG-3′
were used. The product of the second PCR was digested with XbaI and Mph1103I and cloned into the pQβ10 expression vector, which was cleaved by the same restriction enzym...
example 2
Insertion of a Peptide Containing a Lysine Residue into the C / e1 Epitope of HBcAg(1-149).
[0310] The c / e1 epitope (residues 72 to 88) of HBcAg is located in the tip region on the surface of the Hepatitis B virus capsid (HBcAg). A part of this region (Proline 79 and Alanine 80) was genetically replaced by the peptide Gly-Gly-Lys-Gly-Gly (SEQ ID NO: 387) (HBcAg-Lys construct). The introduced Lysine residue contains a reactive amino group in its side chain that can be used for intermolecular chemical crosslinking of HBcAg particles with any antigen containing a free cysteine group.
[0311] HBcAg-Lys DNA, having the amino acid sequence shown in SEQ ID NO:78, was generated by PCRs: The two fragments encoding HBcAg fragments (amino acid residues 1 to 78 and 81 to 149) were amplified separately by PCR. The primers used for these PCRs also introduced a DNA sequence encoding the Gly-Gly-Lys-Gly-Gly peptide (SEQ ID NO: 387). The HBcAg (1 to 78) fragment was amplified from pEco63 using primers ...
example 3
Expression and Purification of HBcAg-Lys
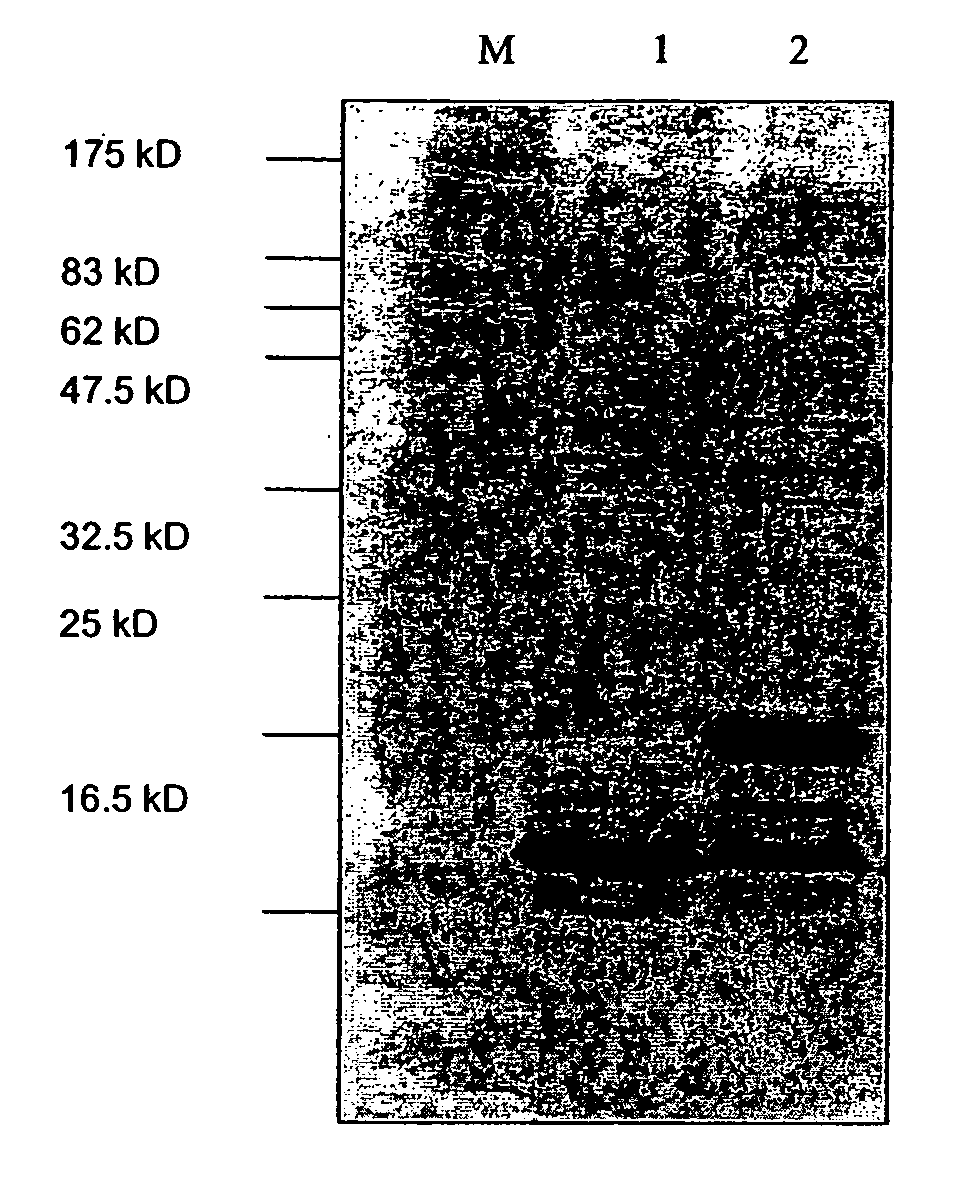
[0314]E. coli strains K802 or JM109 were transformed with pKK-HBcAg-Lys. 1 ml of an overnight culture of bacteria was used to innoculate 100 ml of LB medium containing 100 μg / ml ampicillin. This culture was grown for 4 hours at 37° C. until an OD at 600 nm of approximately 0.8 was reached. Induction of the synthesis of HBcAg-Lys was performed by addition of IPTG to a final concentration of 1 mM. After induction, bacteria were further shaken at 37° C. for 4 hours. Bacteria were harvested by centrifugation at 5000 x g for 15 minutes. The pellet was frozen at -80° C. The pellet was thawed and resuspended in bacteria lysis buffer (10 mM Na2HPO4, pH 7.0, 30 mM NaCl, 0.25% Tween-20, 10 mM EDTA) supplemented with 200 μg / ml lysozyme and 10 μl of Benzonase (Merck). Cells were incubated for 30 minutes at room temperature and disrupted by sonication. E. coli cells harboring pKK-HBcAg-Lys expression plasmid or a control plasmid were used for induction of...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular mass | aaaaa | aaaaa |
| mass | aaaaa | aaaaa |
| molecular mass | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More