Diterpene-Producing Unicellular Organism
a technology of unicellular organisms and diterpenes, applied in the field of molecular genetics of yeast, metabolically engineered yeast, can solve the problems of inability to efficiently and inexpensively produce diterpenes, limited natural sources, and inability to commercialize total syntheses, etc., to achieve the effect of increasing sterol metabolic flux
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Methods and Materials
[0133] Restriction enzymes, DNA polymerase I large (Klenow) fragment, T4 DNA polymerase, calf intestinal alkaline phosphatase, single-stranded binding protein, and M13K07 helper phage were purchased from New England BioLabs. Ligation reactions utilized Fast-Link DNA ligation kit purchased from Epicentre Technologies. Expand high fidelity polymerase kit used for PCR was purchased from Boehringer-Mannheim. Pfu polymerase and pGEM-T vector kit were purchased from Promega. pT7Blue T-vector was purchased from Novagen. Ex-Taq was purchased from Panvera. Zymolyase 100T was purchased from Seikagaku Corporation. Media ingredients were purchased from Fisher Biotech. Reagent chemicals were purchased from Sigma Chemical Company. Organic solvents were purchased from EM Science.
[0134] All E. coli cultures were cultivated in sterile Luria broth, LB. E. coli selective plates, LB-amp, were impregnated with ampicillin (250×stock: 25 mg / mL, filter sterilized) to a final concentr...
example 2
Production of GGPP in De Novosterol Biosynthesis
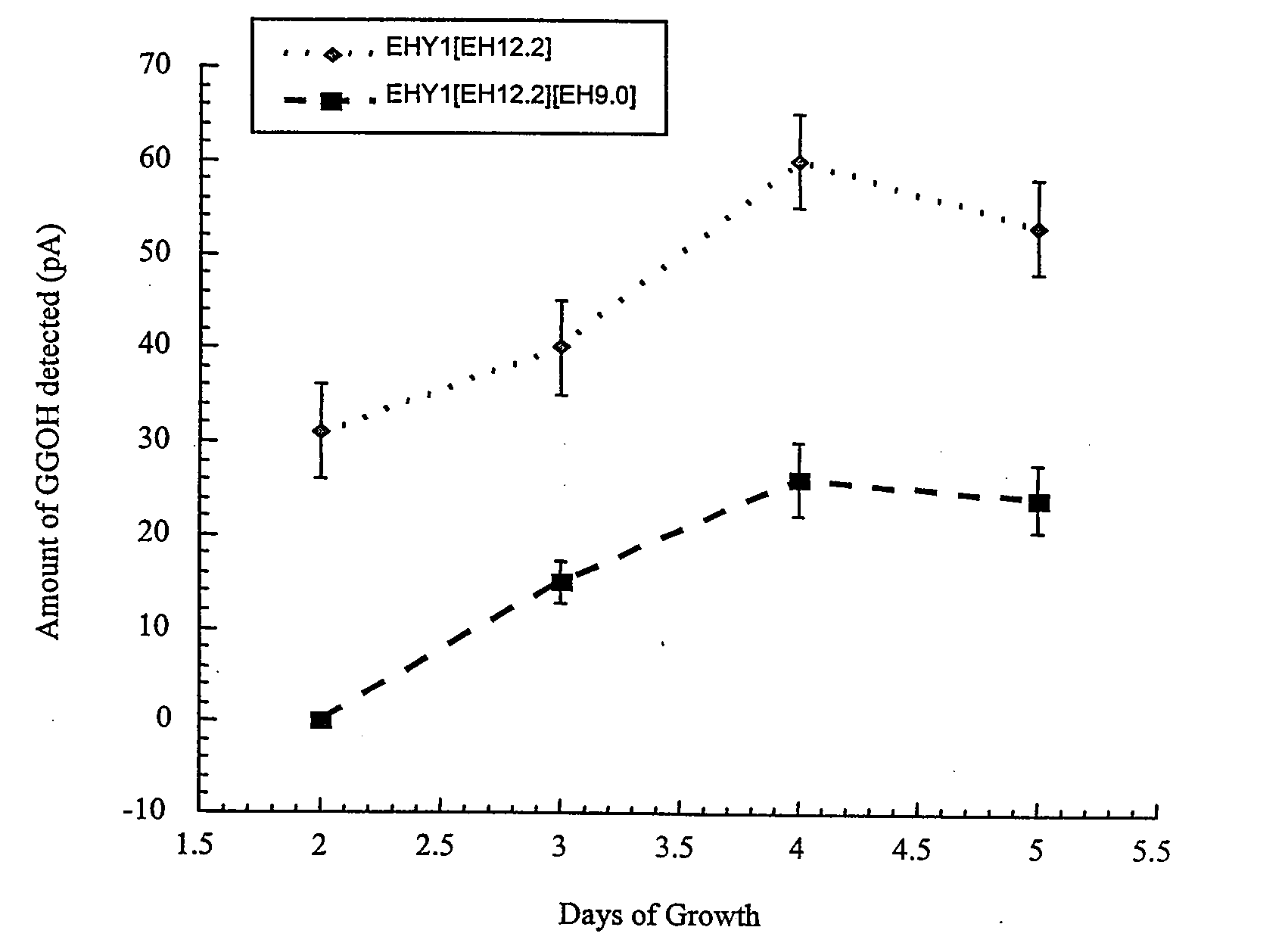
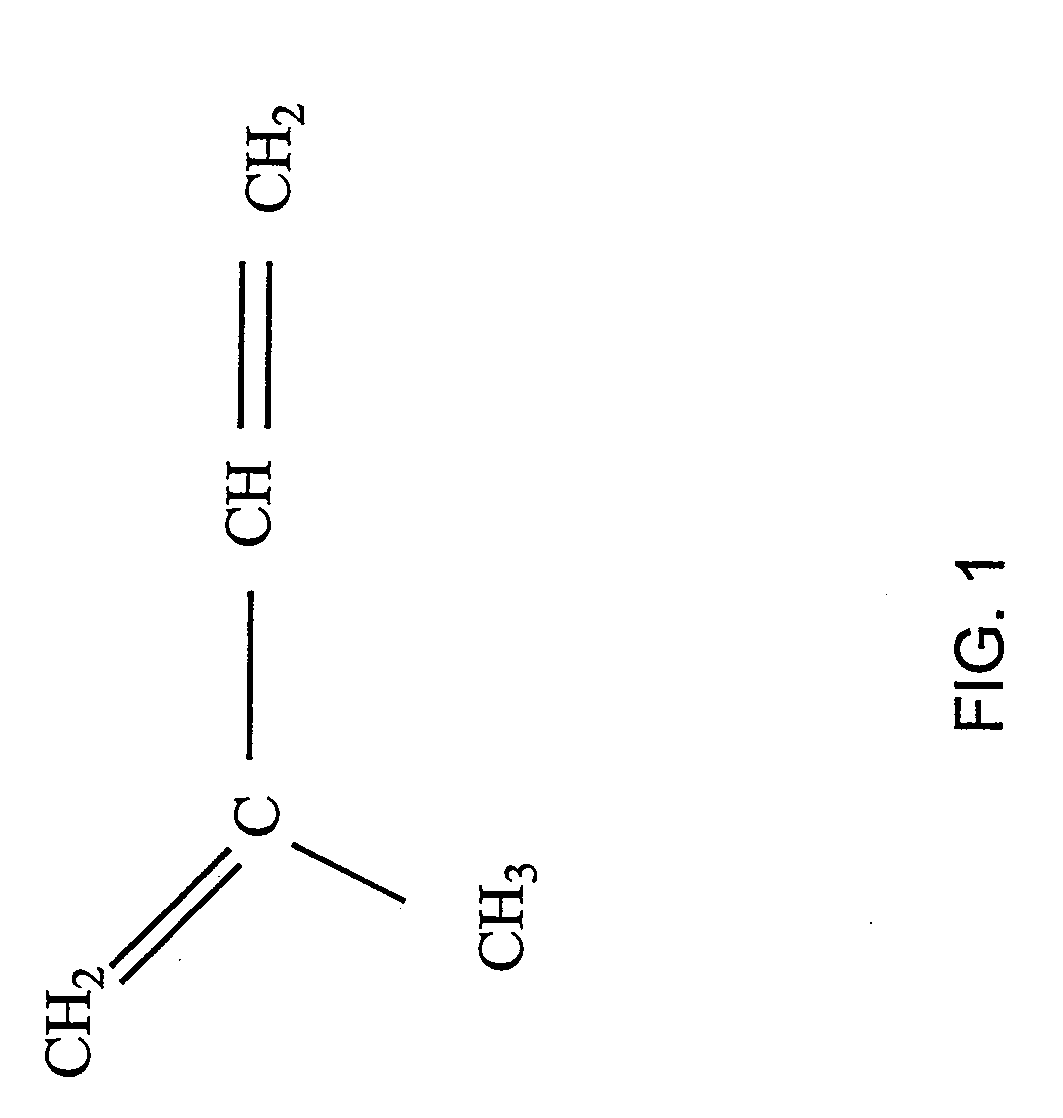
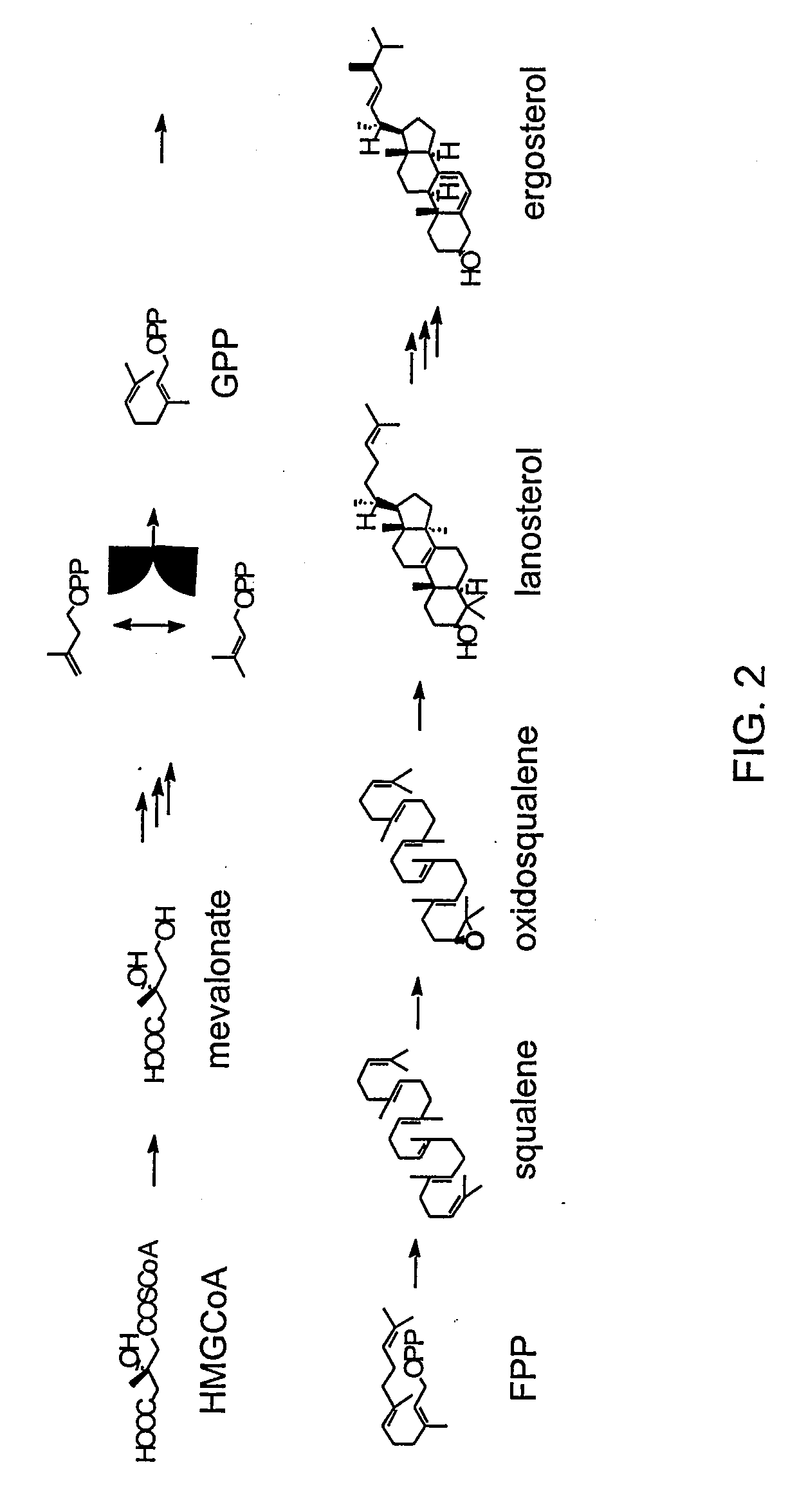
[0136] The amount of endogenous GGPP available in yeast having a native sterol biosynthetic pathway was established. FIG. 2 illustrates the main intermediates of de novo sterol biosynthesis in native yeast. FIG. 3 illustrates the terpene sub-classes and the sterol intermediates that serves as their biosynthetic precursors. Diterpenes are synthesized from (e.g., diterpene precursor) the metabolic intermediate geranylgeranyl pyrophosphate (GGPP).
[0137] Wild type yeast JBY575 (Alani et al., 1987) was transformed with a vector comprising a nucleic acid sequence encoding A. grandis abietadiene synthase (Mende et al., 1997). The culture media contained a polyaromatic resin that indiscriminately adsorbed molecules onto its surface. The induced culture was filtered and extracted to remove the diterpene product, and about 0.01 mg / L abietadiene (extrapolated from the internal standard longifolene at known concentrations) was observed by GC and...
example 3
Generation of GGPP-Synthesizing Yeast
[0138] Investigation of increasing GGPP biosynthesis included heterologous expression of the S. cerevisiae geranylgeranyl diphosphate synthase (BTS1) under transcriptional control of the inducible GAL1 promoter (Jiang et al., 1993).
[0139] The BTS1 nucleic acid sequence was isolated from λ phage received from ATCC using standard methods. Phage DNA containing S. cerevisiae BTS1 was digested with Xho1 and Kpn1 to release a 7 kb DNA fragment that subsequent to purification was ligated into pBluescript (II) KS+ digested with the same two enzymes. Propagation in DH5αyielded pEH1.0. Excess sequence was removed from the insert of pEH1.0 to yield pEH1.1.
[0140] The native promoter of BTS1 was removed by installing a Sal1 site immediately upstream of the start codon by site-directed mutagenesis employing the oligonucleotide sequence GP5S: 5′-TATCTTGGCCTCCATGTCGACTCCAGACTCGTAAAC-3′ (SEQ ID NO:408) and standard methodologies known in the art. The resulting...
PUM
| Property | Measurement | Unit |
|---|---|---|
| dry weight | aaaaa | aaaaa |
| dry weight | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More