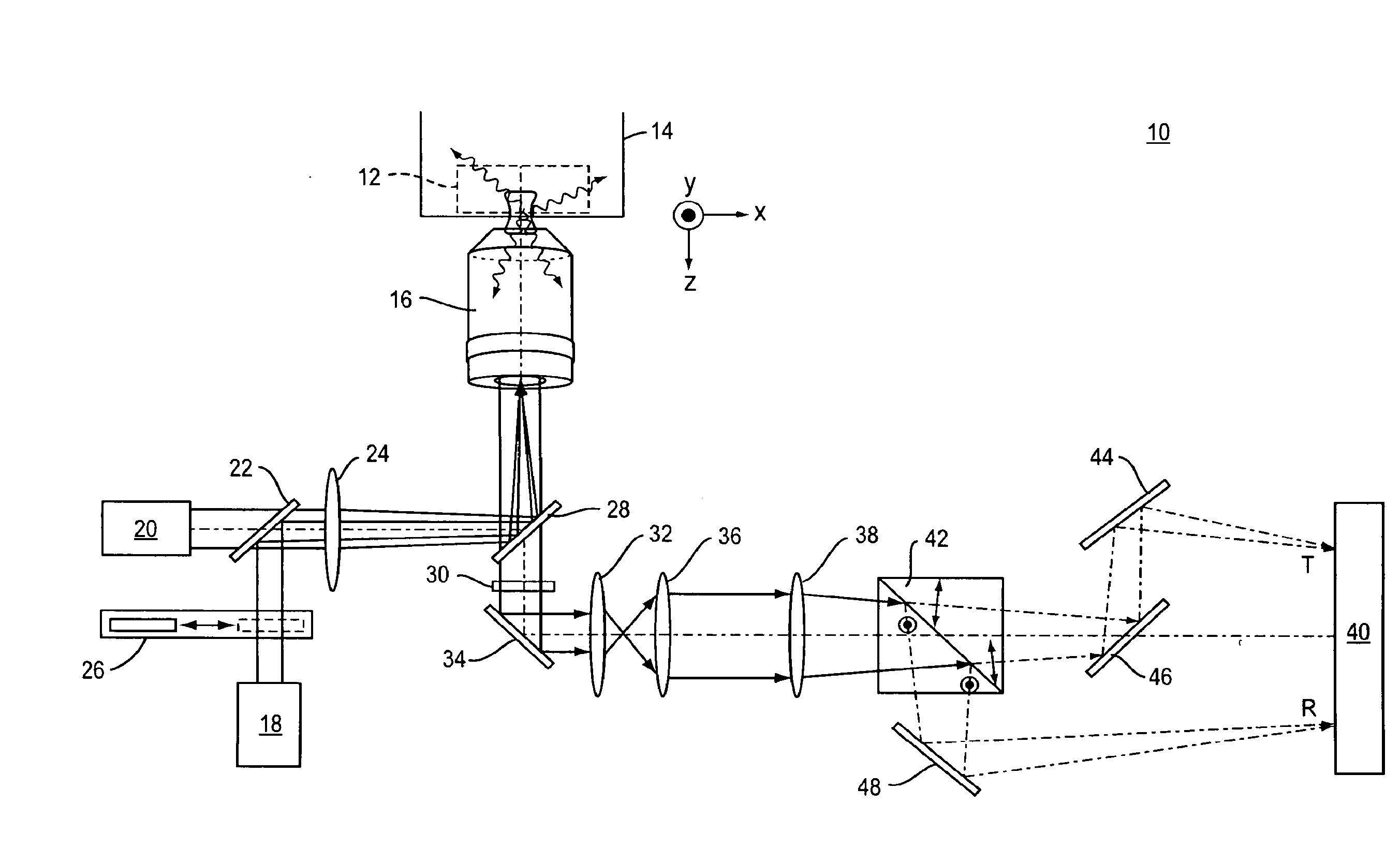
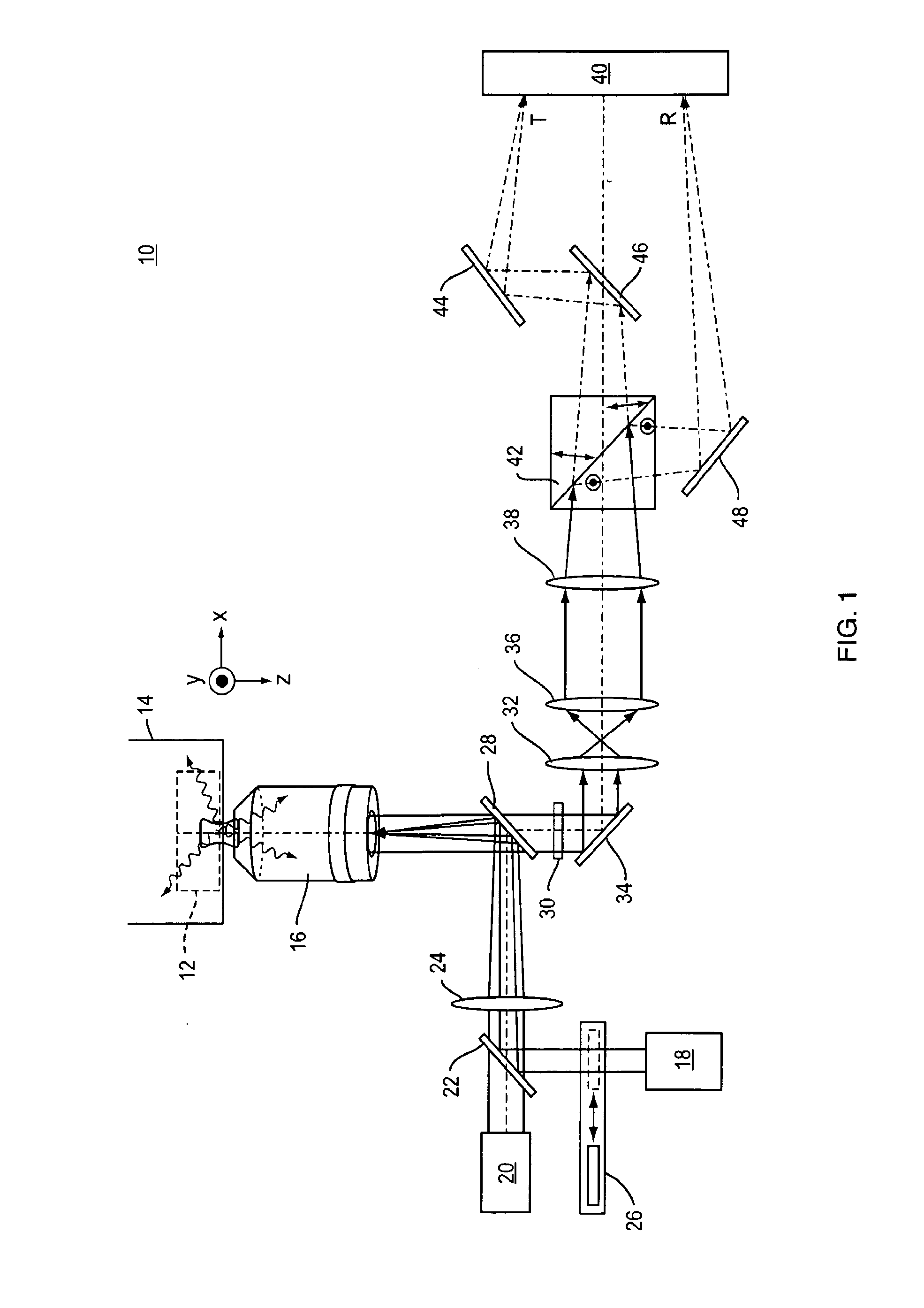
[0008]The system of the present invention incorporates a polarizing
beam splitter into the detection path of a standard FPALM
microscope. This modification allows simultaneous, spatially separate imaging of the fluorescence emitted by a molecule, and this emission is polarized parallel and perpendicular to the excitation polarization. The present invention also modifies the standard FPALM system by adding lenses which expand the emitted fluorescent image paths after polarization, and additional mirrors which adjust the two detection paths to have the same or nearly the same total length from the beam splitter to the
image detector. The method of the present invention analyzes the relative intensities of molecules in the two images to yield the
anisotropy of each localized molecule. Two-dimensional maps (images) of single-molecule anisotropy can be obtained with significantly improved spatial resolution.
[0009]In one example of the system and method of the present invention, the sample is placed on the stage of any suitable
microscope together with a suitable
imaging lens. The use of a water-immersion lens is advantageous because it minimizes aberrations when imaging a sample that is also in water. The sample is illuminated using a
light source of suitable
wavelength. In one embodiment of the present invention the
light source used is a
laser. In another embodiment the
light source of the
microscope system includes two lasers: an activation
laser and a readout
laser of suitable
wavelength. The light source is focused in the objective back-aperture to cause a large area of the sample to be illuminated.
[0010]In one embodiment, illumination using a relatively unfocused
Gaussian beam is advantageous because it reduces the tipping of the polarization toward the z-axis which results from a high-
numerical aperture diffraction-limited focus. In another embodiment, the intensities of the illumination light source are modulated at one or more wavelengths. This modulation can be accomplished using a mechanical or optical
shutter or an electrooptic modulator such as a Pockel'
s cell. This modulation allows sequences of optical pulses to prepare sample molecules in different photophysical states. In yet another embodiment, polarization of the illumination light source is modulated using mechanical or optical shutters which allow illumination light of different polarizations to pass. In embodiments using two lasers as the illumination light source, either the activation or readout beam or both may be modulated in this way. One embodiment includes splitting the illumination light into two or more separate paths with different polarizations that are independently shuttered. Another embodiment modulates the illumination polarization using an electrooptic modulator such as a Pockel'
s cell. This modulation will allow molecules with different orientations to be selectively excited.
[0015]The ability to image anisotropy with resolution below the
diffraction limit presents several interesting opportunities, most importantly the ability to image short-range order and to quantify the degree of preferential orientation of molecules. As long as the limitations of the method are taken into account, we can use the anisotropy to estimate the degree of alignment (but not the precise angle) between the transition-
dipole moment of the emitting molecule (the
fluorophore) and the excitation laser
beam polarization. Interactions between membrane domains and the
cytoskeleton, such as those found in focal adhesions, are expected to result in preferential orientation of molecules, but the size of those structures is generally well below the diffraction limit. The improved resolution in P-FPALM will allow quantification in the order of proteins and lipids in membrane domains at length scales inaccessible to
standard methods.
[0016]In addition to its dramatically improved spatial resolution, P-FPALM provides absolute numbers of molecules and can quantify heterogeneous populations of molecules, both which are inaccessible to conventional methods. P-FPALM provides a means to measure molecular positions and orientations in biological structures in a crucial, but previously inaccessible, range of length scales. Furthermore, P-FPALM will be compatible with live-
cell FPALM, PALM (photo-activated localization microscopy) and
STORM (stochastic
optical reconstruction microscopy) using widefield excitation, and with multi-
color imaging.
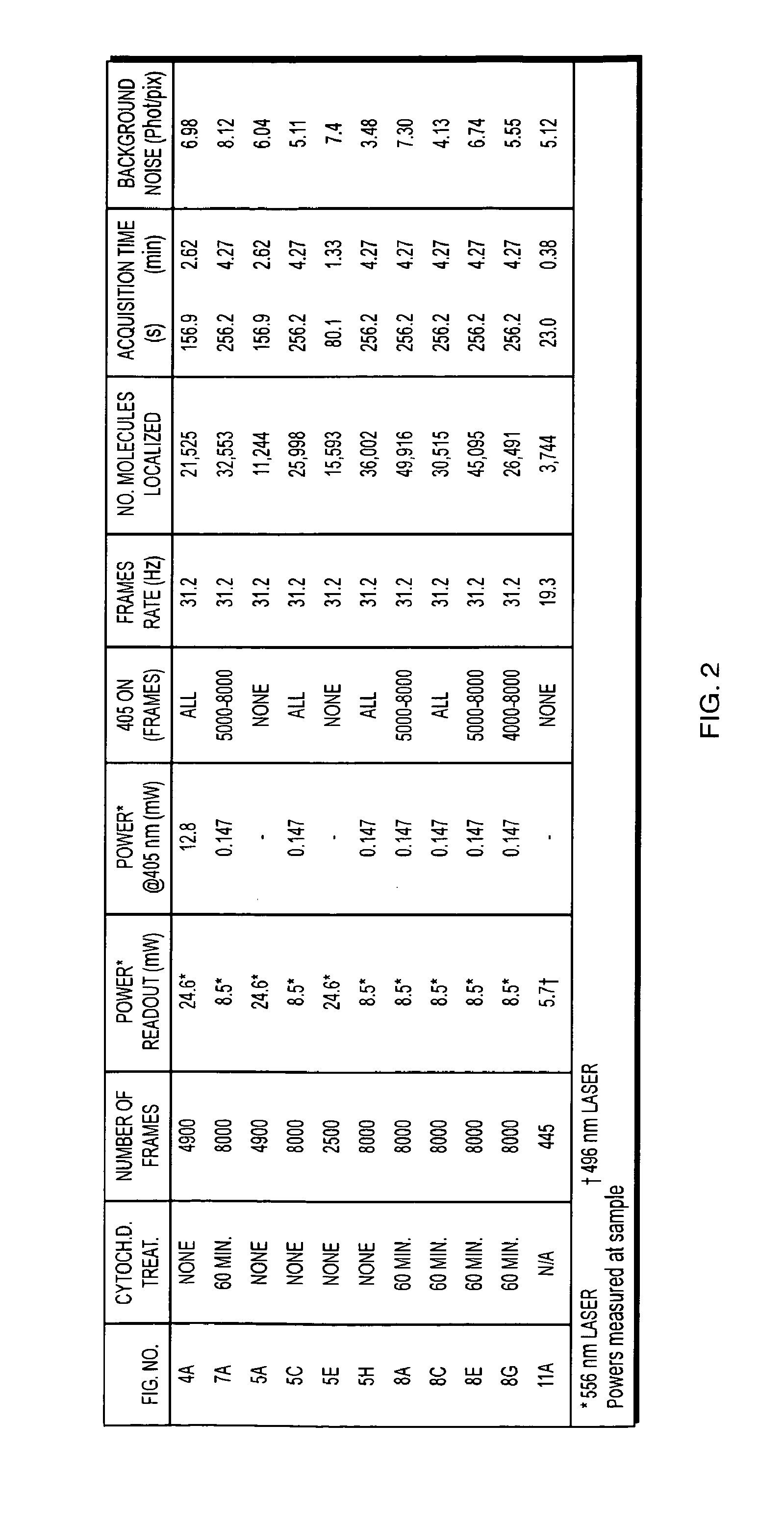
[0019]Longer acquisitions may allow higher molecular densities to be observed in well-immobilized samples. Extension of the technique to three-dimensional (3D) imaging is both possible and useful, considering that structures such as
actin will span many focal planes. Hence, 3D imaging would capture larger numbers of total molecules in different focal planes and allow extended structures to be visualized even more comprehensively.
 Login to View More
Login to View More  Login to View More
Login to View More