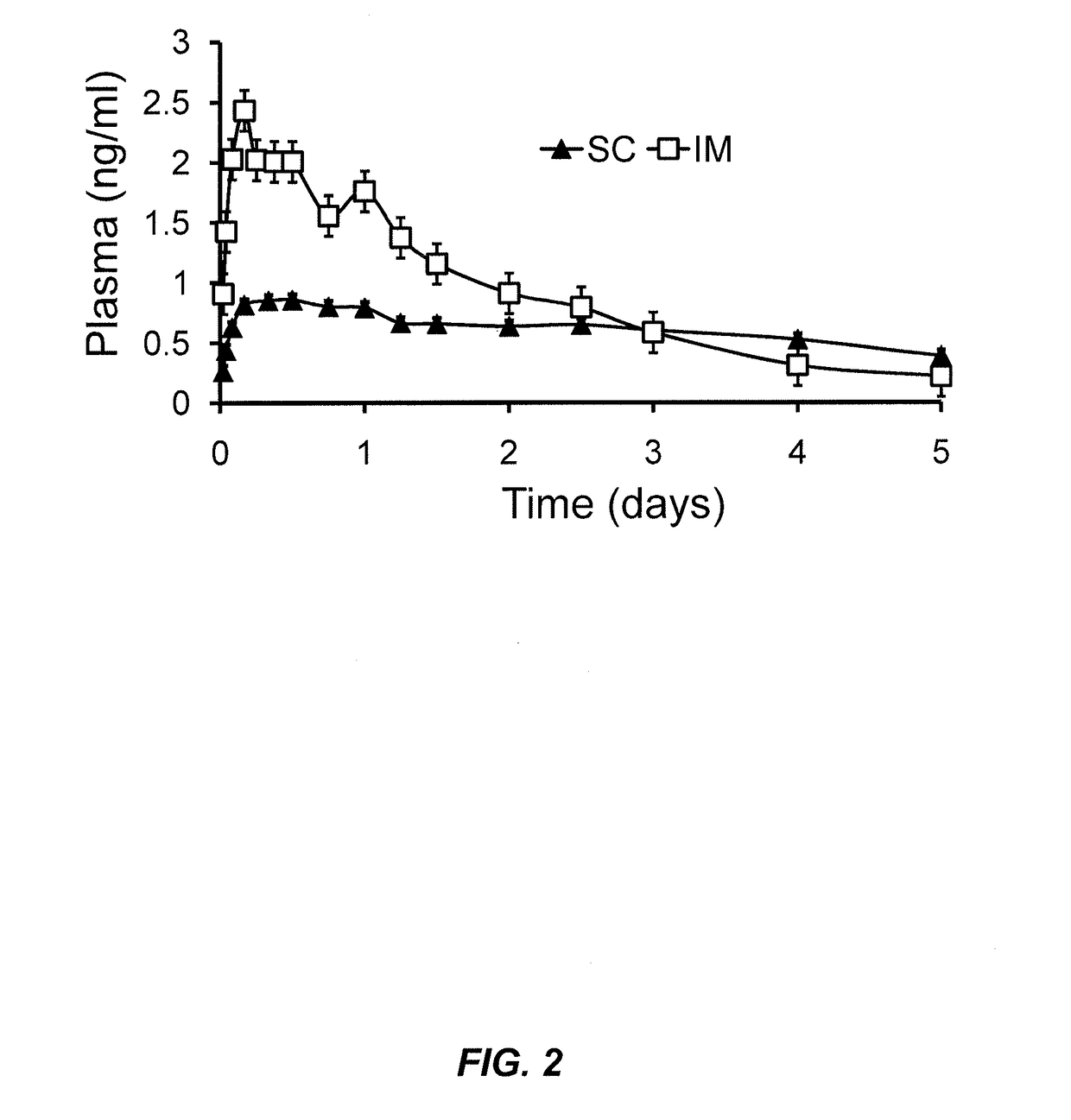
Phospholipid depot
a phospholipid and composition technology, applied in the direction of aerosol delivery, inorganic non-active ingredients, metabolic disorders, etc., can solve the problems of slow release of trapped drugs, large needle diameter, incompatible strong solvents of most biological molecules such as protein drugs, etc., and achieve the effect of convenient injection
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Preparation of Aqueous Gels (Having No Pharmaceutically Active Agent) Using Lecithin
[0208]
ComponentsF-1T-3T-4T-5Compositions(Wt % in primary dispersion)(Wt % in PG)Sesame oil4.06.68.010.1Soy lecithin15.024.730.238.0Cholesterol0.61.01.21.5Vitamin E0.30.50.60.8succinate (VES)Sucrose5.08.210.112.7De-ionized water75.159.050.037.0(DI-water)Total100100.0100.0100.0NameSupplierGradeSesame oilCrodaSuper-refined, USPSoy lecithinLIPOIDEP(Phospholipon 90G)CholesterolSolvay PharmaceuticalsHP, NFVESSpectrum Chem.USPSucroseSpectrum Chem.NFDe-ionized waterLatitude Pharmaceuticals Inc.
Procedure
[0209]The F-1 primary dispersion first was prepared and converted to three aqueous gels (T-3, T-4 and T-5) by the following procedure:[0210]1. Weigh out sesame oil, Phospholipon 90 G and cholesterol into a glass flask.[0211]2. Add 50 mL ethanol USP.[0212]3. Rotate the flask to dissolve all solids.[0213]4. Vacuum dry to remove ethanol to less than 1% by weight.[0214]5. Add VES (in a 5% stock solution), sucrose ...
example 2
Preparation of Aqueous Gels Containing Recombinant Human Insulin for Basal Insulin Therapy
[0221]Aqueous gels containing recombinant human insulin and 40%, 50% and 60% water were prepared using lecithin. Insulin was introduced into the process before microfluidization and the resulting nanodispersion was filtered for sterilization. Thus, no heat or radiation sterilization was needed in the process.
Compositions (% wt)NanodispersioAqueous GelComponentF-1F-2F-3F-4Recombinant74.9 (IU / g)84.9 (IU / g)100 (IU / g)115.4 (IU / g)human insulinSesame oil4.06.438.039.64Soy lecithin15.024.1030.1236.14Cholesterol0.60.961.201.45Vitamin E0.30.480.600.72succinate(VES)Sucrose5.08.0310.0412.05EDTA0.0150.0180.0200.023disodiumdehydrateDe-ionized75.1605040water(DI-water)
[0222]Procedure
[0223]The F-1 nanodispersion was first prepared and converted to three anhydrous gels (F-2, F-3 and F-4) containing 40, 50 and 60% water as follows:[0224]1. Weigh out sesame oil, soy lecithin and cholesterol into a glass flask.[02...
example 3
Preparation of Aqueous Gels Containing Recombinant Human Insulin for Basal Insulin Therapy
[0236]The procedure was directed to preparation of aqueous gels containing recombinant human insulin at 100 IU / mL and 50% water using a soy lecithin (F-1G) and a synthetic phospholipid (F-5G). The insulin was introduced into the process before microfluidization.
ComponentF-1GF-5GCompositions (% wt)(Nanodispersion)(Gel)(Nanodispersion)(Gel)Recombinant human insulin0.2840.379*0.2840.379*Sesame oil48.0348.03Phospholipon 90G (PL90G)1530.121-Palmitoy1-2-01eoyl-sn-glycero-3-1530.12phosphocholine (POPC)Cholesterol0.61.20.61.2Vitamin E succinate (VES)0.30.60.30.6Sucrose510.04510.04EDTA disodium dehydrate0.0110.0150.0110.015Histidine0.0780.1040.0780.104De-ionized water (DI-water)75.15075.150*Equivalent to 100 IU / g
[0237]Procedure
[0238]The nanodispersions were first prepared and converted to the anhydrous gels following the steps below:[0239]1. Weigh out sesame oil, PL9OG or POPC and cholesterol into a gla...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com