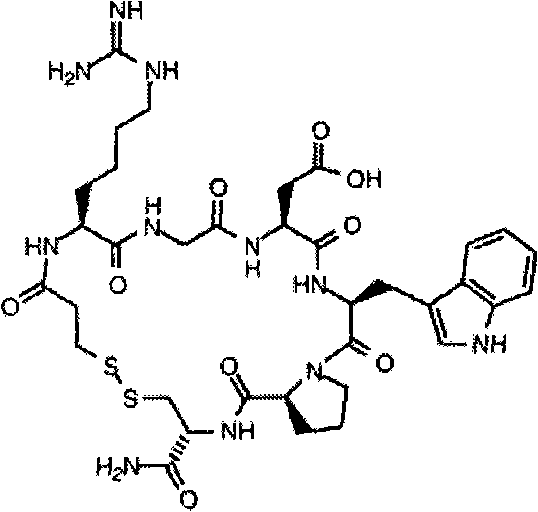
Eptifibatide powder injection
A technology of tebatide powder and eptifibatide, which is applied in the direction of cyclic peptide components, powder delivery, medical preparations of non-active ingredients, etc., can solve the problems of difficulty, cost increase, high freeze-drying, etc., and achieve transportation and storage Convenience, reduce production cost, improve stability
Inactive Publication Date: 2010-02-03
NANJING CHANGAO PHARMA SCI & TECH CO LTD
View PDF0 Cites 2 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
The solubility of Efibuta in water is low. When the pH value reaches 7, it is easy to precipitate out of the solution. The general co-solvent has little effect on its solubility, and has certain viscosity and aggregation. Therefore, the maximum concentration of the infusion solution is generally 20mg / ml, so that the volume of the same dose of efibat injection for each patient will be very large, so that even if a large dose bottle of 100ml: 200mg is given, more than 2 bottles will be needed. Need to occupy a larger space, and the cost will increase accordingly
In addition, efibat has poor stability in aqueous solution, and when the pH value is lower than 4, the related substances increase significantly
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0026] Embodiment 1 20mg specification formula
[0027] Eptifibatide 20.0g
[0028] Citric acid 25.0g
[0029] Lactose 50.0g
[0030] 1MNaOH appropriate amount
[0031] Water for injection to 1500ml
[0032] Makes 1000 bottles
Embodiment 2
[0033] Embodiment 2 20mg specification formula
[0034] Eptifibatide 20.0g
[0035] Citric acid 28.0g
[0036] Lactic acid 2.0g
[0037] Lactose 50.0g
[0038] Mannitol 50.0g
[0039] 1MNaOH appropriate amount
[0040] Water for injection to 1500ml
[0041] Makes 1000 bottles
Embodiment 3
[0042] Embodiment 3 20mg specification formula
[0043] Eptifibatide 20.0g
[0044] Sodium citrate 30.0g
[0045] Mannitol 120.0g
[0046] 1M HCl qs
[0047] Water for injection to 2000ml
[0048] Makes 1000 bottles
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention belongs to the field of medicinal preparations and relates to an eptifibatide powder injection, which mainly comprises eptifibatide, carbohydrates and an acidity regulator. The eptifibatide powder injection has the characteristics of high concentration and good stability, can be used for treating acute coronary syndrome including unstable angina pectoris and non-Q wave infarction andcan also be used in percutaneous coronary intervention including angioplasty and atherectomy.
Description
technical field [0001] The invention belongs to the field of pharmaceutical preparations and relates to eptifibatide powder injection. Background technique [0002] Eptifibatide (Efibate) is a glycoprotein II b / IIIa receptor antagonist, which binds to the GPIIb / IIIa receptor on the platelet membrane and occupies the binding site on it, allowing the platelet GPIIb / IIIa receptor The body cannot bind to fibrinogen, thereby inhibiting platelet aggregation. Clinically indicated for the treatment of acute coronary syndromes, including unstable angina or non-Q-wave myocardial infarction (ie, persistent episodes of chest pain within 24 hours, ECG changes, and / or elevated cardiovascular system enzymes); also for percutaneous coronary use Interventional therapy, including angioplasty or atherectomy. In July 1998, it was first listed in the United States under the trade name Intrifiban by Schering-Plough Company, and in 1999 it was launched in Europe under the trade name Intrifiban. ...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): A61K9/19A61K38/12A61K47/26A61K47/10A61K47/12A61P9/10
Inventor 谢晓燕张自强黄海燕
Owner NANJING CHANGAO PHARMA SCI & TECH CO LTD