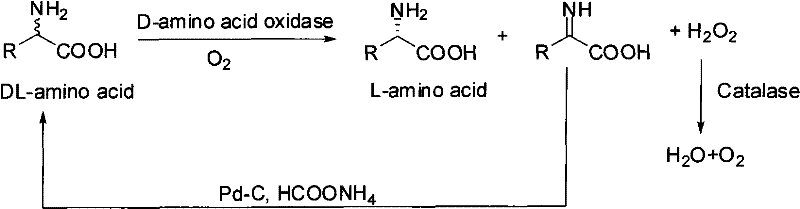
Bio-catalytic deracemization preparation method of non-natural L-amino acid
A technology of biocatalysis and biocatalyst, which is applied in the field of preparation of chiral pharmaceutical intermediates by biocatalytic asymmetric oxidation, can solve the problems of sensitivity, easy decomposition, unreported non-natural L-amino acid method, etc., and achieve simple process and environmental friendliness , good versatility
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0021] Example 1: L-2-aminobutyric acid
[0022] 2.06g (0.02mol) DL-2-aminobutyric acid was dissolved in 50ml ammonium formate (1.0mol / L, adjust the pH to 7.2 with ammonia water) solution, add 2.06g immobilized D-amino acid oxidase (58U / g), 1ml of catalase solution (50000U / ml), 1g of Pd-C catalyst (10%), continuously fed with oxygen, stirred and reacted at 30°C for 16h, the optical rotation of the reaction solution no longer increased. Remove the immobilized D-amino acid oxidase and Pd-C by filtration, concentrate the filtrate to about 10ml, adjust the isoelectric point, freeze and precipitate white crystals, and dry to obtain 1.8gL-2-aminobutyric acid, the yield is 87%, ee>99 %.
Embodiment 2
[0023] Example 2: L-2-aminovaleric acid
[0024] 2.90g (0.025mol) of DL-2-aminovaleric acid was dissolved in 50ml of ammonium formate (1.0mol / L, the pH was adjusted to 7.2 with ammonia water), and 2.90g of immobilized D-amino acid oxidase (58U / g) was added, 1.5ml of catalase solution (50000U / ml), 1g of Pd-C catalyst (10%), continuously fed with oxygen, stirred and reacted at 30°C for 16h, the optical rotation of the reaction solution no longer increased. Remove the immobilized D-amino acid oxidase and Pd-C by filtration, concentrate the filtrate to about 10ml, adjust the isoelectric point, freeze and precipitate white crystals, and dry to obtain 2.6gL-2-aminovaleric acid, the yield is 90%, ee>99 %.
Embodiment 3
[0025] Example 3: L-2-aminoadipic acid
[0026]3.22g (0.02mol) DL-2-amino adipic acid was dissolved in 50ml ammonium formate (1.0mol / L, adjust the pH to 7.2 with ammonia water) solution, add 3.22g immobilized D-amino acid oxidase (58U / g) , 1ml catalase solution (50000U / ml), 1g Pd-C catalyst (10%), continuously feed oxygen, stir and react at 30°C for 20h, and the optical rotation of the reaction solution no longer increases. Remove the immobilized D-amino acid oxidase and Pd-C by filtration, concentrate the filtrate to about 10ml, adjust the isoelectric point, freeze and precipitate white crystals, and dry to obtain 2.8g of L-2-aminoadipic acid with a yield of 87%, ee> 99%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More