Quality control method of granules capable of strengthening and consolidating body resistance
A quality control method and a technology for strengthening the body and strengthening the foundation, which are applied in the fields of chemotherapy combined drugs and cancer patient radiotherapy, which can solve the problems of no chemical identification of the constituent drugs and a small number of quantitative determinations, and achieve strong specificity, easy operation and high precision. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
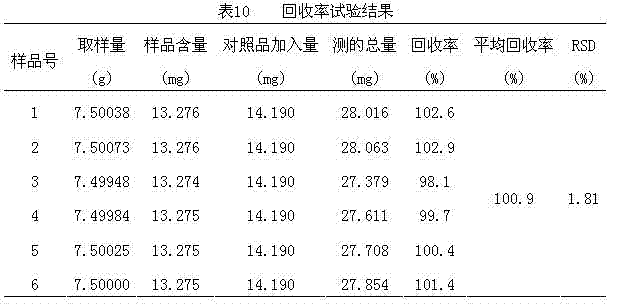
Image
Examples
Embodiment 1
[0036] Example 1: Preparation of Fuzheng Guben Granules.
[0037] Prescription: Skullcap 1667g, Ligustrum lucidum 1667g, Epimedium 1333g, Polygonum multiflorum 1333g, Rehmannia glutinosa 1333g, Polygonatum 1333g, Rubia 1000g, Ginseng 333g.
[0038] Preparation method: crush the above eight flavors and ginseng into coarse powder, add water to decoct twice, each time for 2 hours, combine the decoction, and filter; add the remaining seven flavors such as medicinal dregs and scutellaria baicalensis, add water to decoct three times, each time for 1.5 hours, combine Decoction, filter, combine the filtrate with the above ginseng extract, concentrate to a clear paste with a relative density of about 1.35 (60°C), add 3 parts of sucrose, 1 part of dextrin and an appropriate amount of ethanol, make granules, dry, and make 1000 bags, that is.
[0039] Properties: This product is brown-yellow granules; sweet and slightly bitter.
[0040] Functions and indications: nourishing qi and nouri...
Embodiment 2
[0044] Example 2: TLC identification of Scutellaria baicalensis.
[0045] Get 15g of the medicine of Example 1, grind finely, add methanol 20ml, shake for 30 minutes, filter, and the filtrate is used as need testing solution. Take another baicalin reference substance, add methanol to make a solution containing 1mg per 1ml, as the reference substance solution. According to the thin-layer chromatography (Appendix VI B of the Chinese Pharmacopoeia 2010 edition), draw 5 μl of the test solution and the reference solution, respectively, and place them on the same silica gel G thin-layer plate, and use ethyl acetate-butanone-formic acid - The upper solution of water (5:3:1:1) is the developer, develop, take out, dry in the air, and spray with 1% ferric chloride ethanol solution. In the chromatogram of the test product, there are spots of the same color at the position corresponding to the chromatogram of the reference product.
Embodiment 3
[0046] Example 3: TLC identification of Ligustrum lucidum.
[0047] Take 10 g of the drug of Example 1, grind it finely, add 40 ml of ether, reflux for 1 hour at low temperature, filter, evaporate the filtrate to dryness, add 1 ml of ethanol to the residue to dissolve, and use it as the test solution. Take another oleanolic acid reference substance, add ethanol to make 1ml solution containing 1mg, as the reference substance solution. According to the thin-layer chromatography (Appendix VI B of the Chinese Pharmacopoeia 2010 edition), draw 15 μl of the test solution and 5 μl of the reference solution, respectively spot on the same silica gel G thin-layer plate, and mix with cyclohexane-acetone-ethyl acetate Ester (5:2:1) is used as a developing agent, develop, take out, dry in the air, spray with 10% sulfuric acid ethanol solution, and heat at 105°C until the spots are clearly colored. In the chromatogram of the test product, there are spots of the same color at the position c...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More