Quality control method for Qiqing toxin-removing particles
A technology of particle quality and control method, applied in measuring devices, instruments, scientific instruments, etc., can solve the problems of complex ingredients, difficult to control content, influence on the safety and effectiveness of traditional Chinese medicine, and achieve the effect of simple operation and improved quality level.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
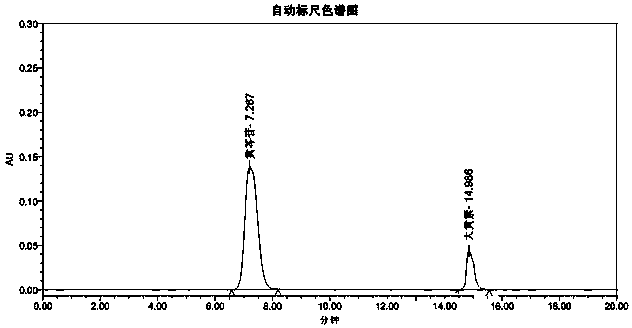
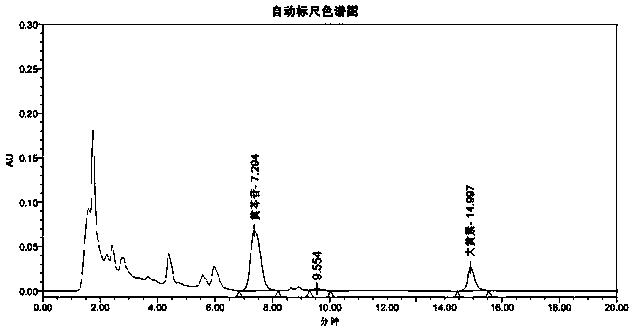
[0024] Embodiment 1HPLC method simultaneously measures the content of baicalin and emodin in Qiqingbaidu granule
[0025] In the embodiment of the present invention, HPLC chromatographic condition is:
[0026] Chromatographic column: Agilent-C18 (150mm X 4.6mm, 5um).
[0027] Mobile phase: acetonitrile-water-phosphoric acid (22:78:0.2)
[0028] Flow rate: 1ml / min; detection wavelength: 270nm; column temperature: 35°C.
[0029] Preparation of reference substance solution: Take the appropriate amount of baicalin reference substance and emodin reference substance that were dried under reduced pressure at 60°C for 4 hours, weigh them accurately, put them in the same 25ml measuring bottle, add 70% ethanol to dissolve and dilute to the mark, shake well , make every milliliter contain the reference substance solution of baicalin 0.3mg, emodin 20ug.
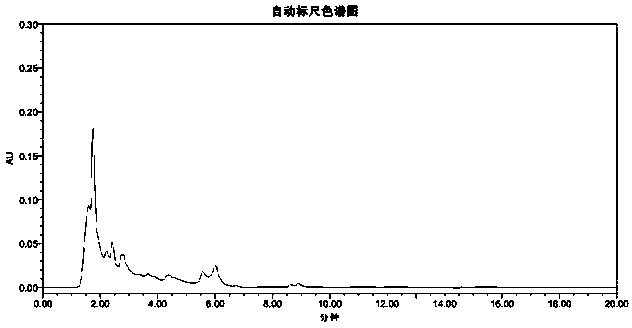
[0030] Preparation of the test solution: Take about 0.2 g of the finely ground test sample, weigh it accurately, put it in a 25ml me...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com