A kind of donepezil hydrochloride crystal form compound and preparation method thereof
A technology of donepezil hydrochloride and its compound, which is applied in the field of medicine, can solve problems such as poor drug stability, and achieve the effects of good stability, high dissolution rate and bioavailability, and simple preparation method
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0028] Take 15 g of donepezil hydrochloride in a reaction flask, add 300 ml of a mixed solution of dimethyl sulfoxide and ethanol (the volume ratio of dimethyl sulfoxide and ethanol is 5:1), heat to 40 ° C, stir to dissolve, and filter while hot ;While stirring, the temperature was lowered to 20°C (the cooling rate was 5°C per minute), and the pre-cooled mixed solvent B was added to the solution at a flow rate of 1.0mL / min (the volume ratio of ethanol and dichloromethane was 1:3) 600ml until the crystals appear, continue to cool down to -5°C (the cooling rate is 1°C per minute), and stir for 3h. After vacuum filtration, the filter cake was vacuum-dried at 50° C. for 6 h to obtain 13.0 g of white needle-like solid, with a yield of 86.3%.
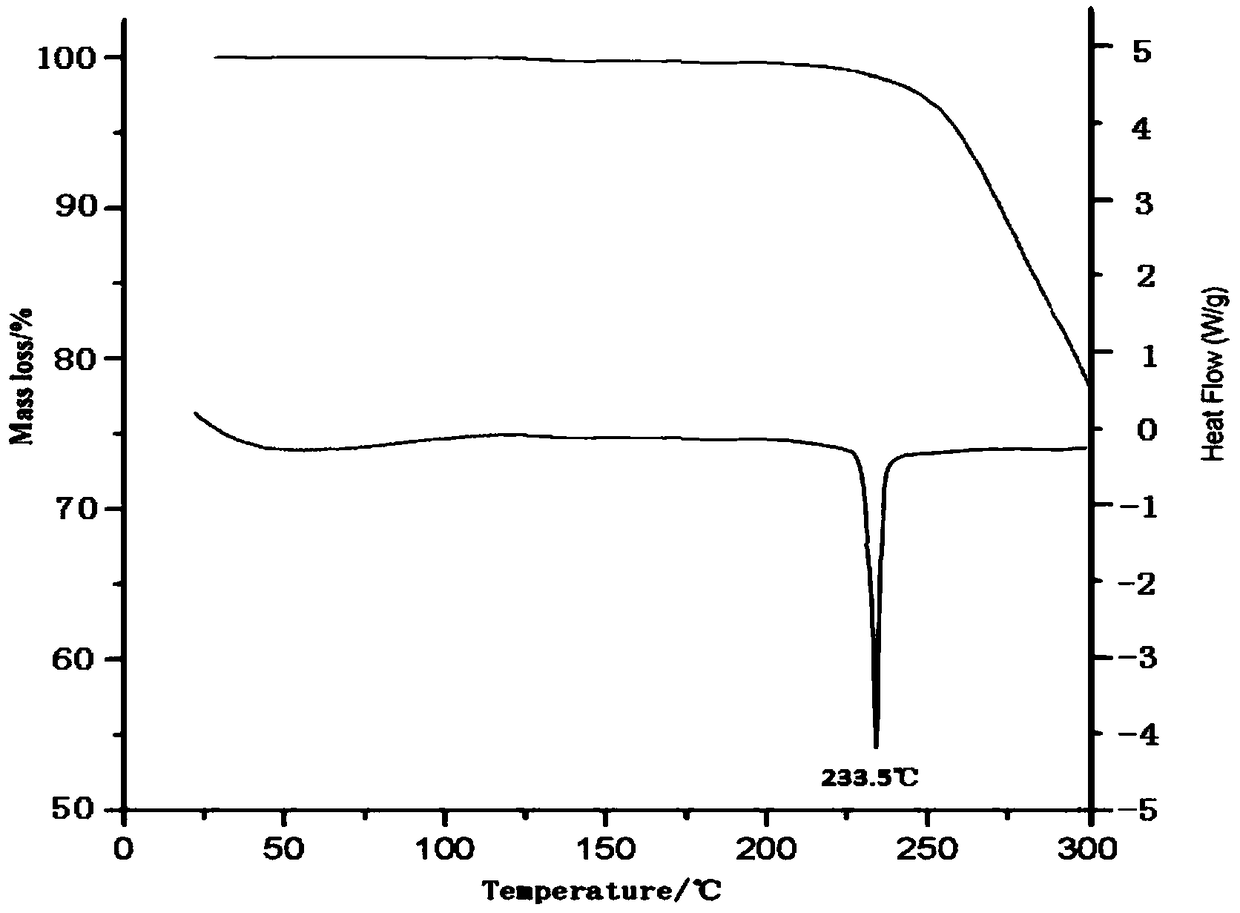
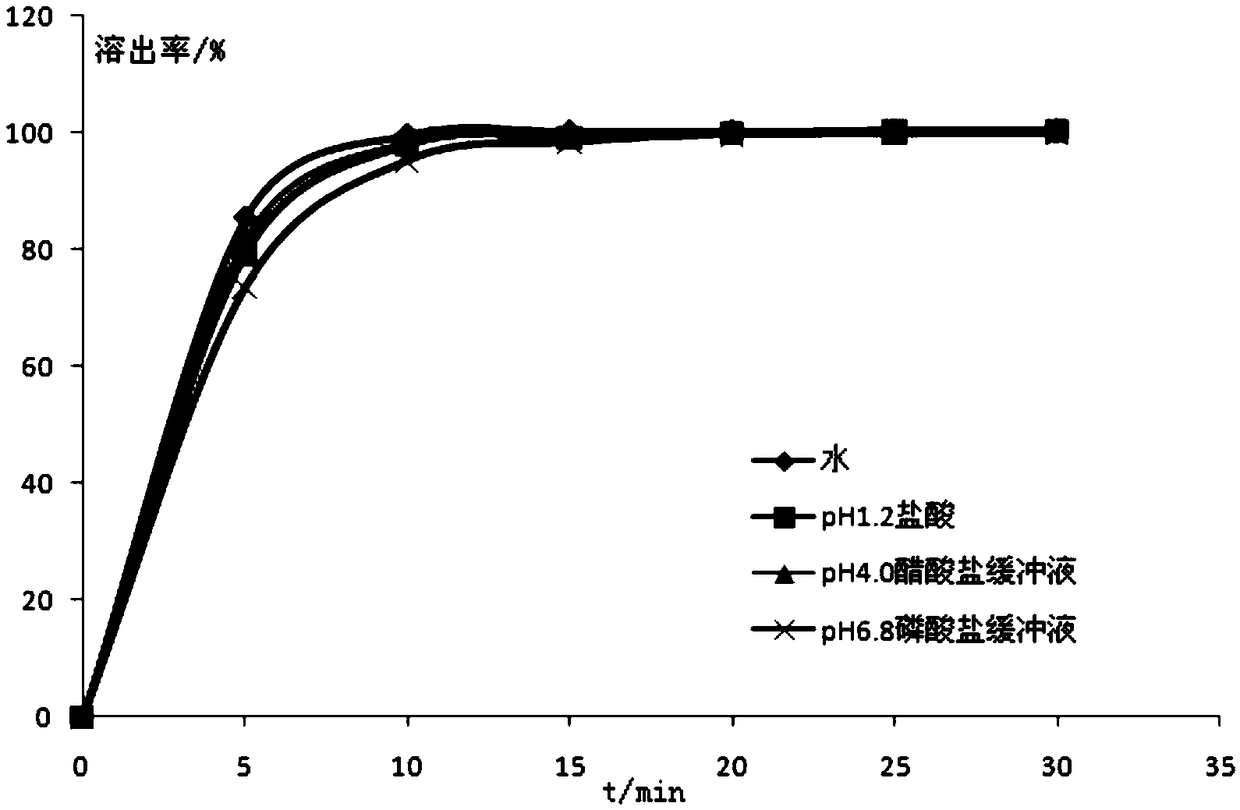
[0029] Related analysis tests:
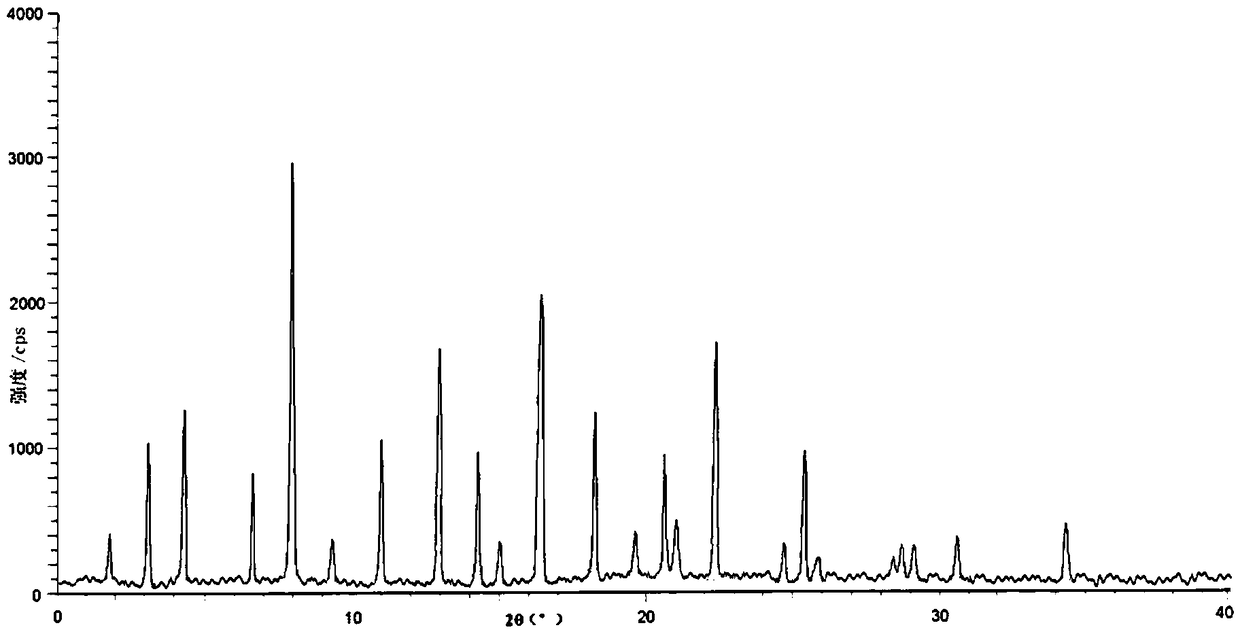
[0030] 1. X-ray powder diffraction test:
[0031] Instrument model: BrukerD8ADVANCE; light source Cu-Ka40kV40mA; graphite monochromator; divergence slit (DS): 1°; LynxEye array detector, scanning mode: θ / θ, ...
Embodiment 2
[0036] Take 10 g of donepezil hydrochloride in a reaction flask, add 100 ml of a mixed solution of dimethyl sulfoxide and ethanol (the volume ratio of dimethyl sulfoxide and ethanol is 6:1), heat to 50 ° C, stir to dissolve, and filter while hot ;While stirring, the temperature was lowered to 25°C (the cooling rate was 5°C per minute), and the pre-cooled mixed solvent B was added to the solution at a flow rate of 1.5mL / min (the volume ratio of ethanol and dichloromethane was 1:3) 300ml until the crystals appear, continue to cool down to -10°C (the cooling rate is 1°C per minute), and stir for 2h. After vacuum filtration, the filter cake was vacuum-dried at 50° C. for 4 hours to obtain 9.06 g of white needle-like solid, with a yield of 90.6%. The X-ray powder diffraction spectrum of the obtained crystal measured by Cu-Kα rays is similar to that of Example 1.
Embodiment 3
[0038] Take 15g of donepezil hydrochloride in a reaction flask, add 225ml of a mixed solution of dimethyl sulfoxide and ethanol (the volume ratio of dimethyl sulfoxide and ethanol is 4:1), heat to 45°C, stir to dissolve, and filter while hot ;While stirring, the temperature is lowered to 30°C (the cooling range is 5°C per minute), and 1125ml of pre-cooled mixed solvent B (the volume ratio of ethanol and dichloromethane is 1:3) is added to the solution at a flow rate of 2mL / min Until the crystals appear, continue to cool down to -5°C (the cooling rate is 1°C per minute), and stir for 2h. After vacuum filtration, the filter cake was vacuum-dried at 50° C. for 5 h to obtain 13.7 g of white needle-like solid, with a yield of 91.5%. The X-ray powder diffraction spectrum obtained by measuring the prepared donepezil hydrochloride crystals using Cu-Kα rays is similar to that of Example 1.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More