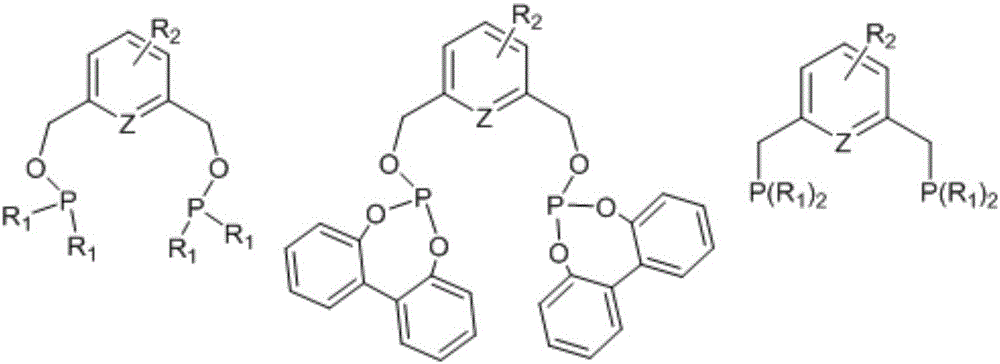
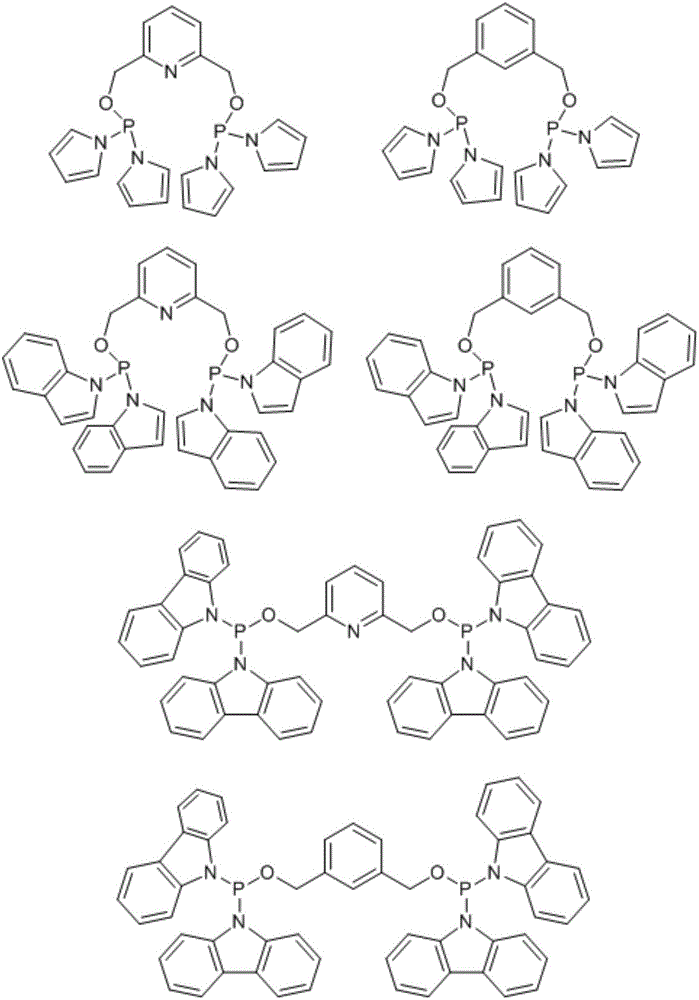
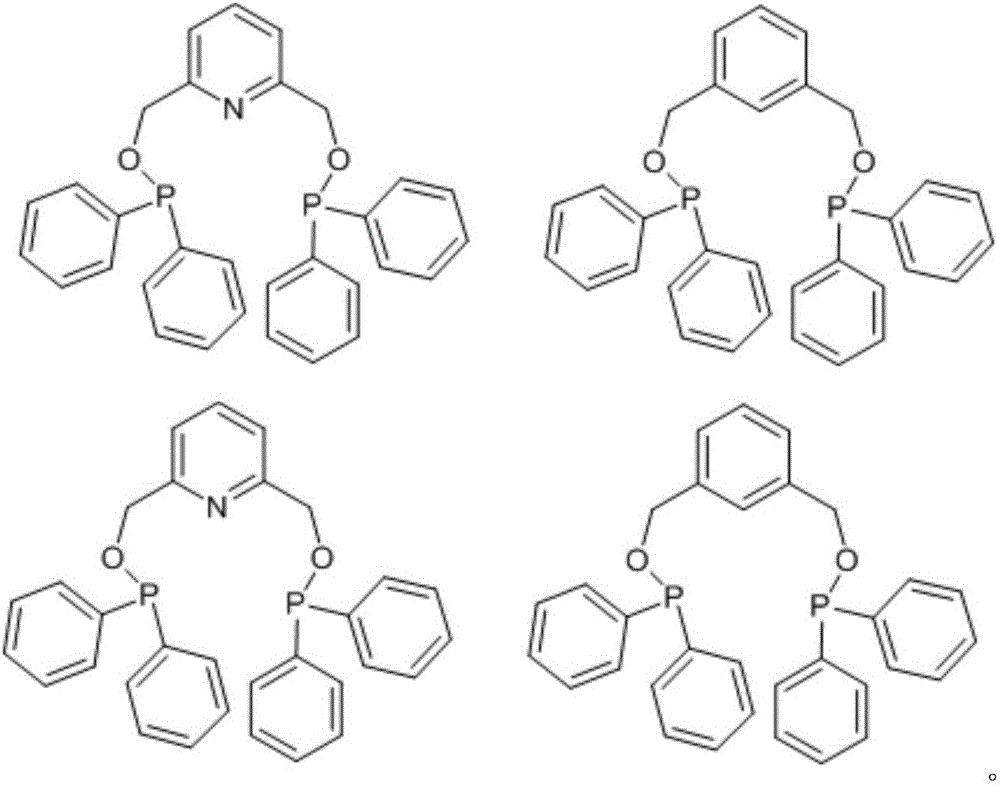
Catalyst for nonterminal olefin hydroformylation reaction and preparation method and application of catalyst
A technology of hydrocarbon hydroformyl and catalyst, which is applied in the field of catalyst preparation and application, can solve few problems, and achieve the effect of improving reaction activity and good catalytic activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0032] Synthesis of 2,2-dihydroxymethylpyridine skeleton:
[0033]
[0034] 2,6-Ethylpyridine dicarboxylate Depend on 2,6-dicarboxylic acid pyridine esterification: add 2,6-dicarboxylic acid pyridine (28g, 168mmol) in a 500mL three-necked flask, then add 300mL ethanol to dissolve the solid, and slowly add acetyl chloride dropwise at room temperature ( 15mL, 212mmol), after the dropwise addition was completed, the reaction temperature was controlled to be less than 30°C. After 24 hours of reaction, the solvent was removed under reduced pressure, 30mL of deionized water was added, the pH was adjusted to 7 with sodium carbonate, and extracted three times with 50mL of ether, combined The extract was dried by adding anhydrous magnesium sulfate for 2 hours, filtered, and the ether was removed under reduced pressure to obtain a white solid, which was dried in vacuo to obtain 30 g of the product with a yield of 80.0% and a melting point of 42-43°C.
[0035] Add in a 500ml three...
Embodiment 2
[0037] Synthesis of pincer-like phosphonamidite bisphosphine ligands:
[0038]
[0039] Dipyrrole phosphorus chloride Synthesized from pyrrole and phosphorus trichloride: in N 2 Or under the protection of Ar gas atmosphere, add anhydrous tetrahydrofuran (120ml) and phosphorus trichloride (5.3ml, 0.06mol) to the 250ml three-necked flask, and add pyrrole (8.4ml, 0.12mol) and trichloride dropwise under ice bath conditions. Ethylamine (25.0ml, 0.18mol) in anhydrous tetrahydrofuran (30ml), slowly added dropwise (about 1h), then warmed up to room temperature and stirred overnight; stop the reaction and let stand for 20 minutes, filter under N2 atmosphere to remove triethylamine Hydrochloride, most of the solvent tetrahydrofuran was removed under reduced pressure, and the residue was distilled under reduced pressure to collect the product at 80°C (0.1mmHg). The product is a colorless oil. Weight 5.3g, yield 45.0%.
[0040] 31 P NMR (166MHz, CDCl3): δ=103.63.
[0041] In a 1...
Embodiment 3
[0045] Synthesis of bidentate phosphonamidophosphine ligands:
[0046]
[0047] diindophos chloride Synthesized from phosphorus trichloride and indole: in N 2 Or under an Ar gas atmosphere, add anhydrous tetrahydrofuran (200ml) and phosphorus trichloride (4.4ml, 0.05mol) respectively to a 500ml three-necked flask, and add indole (11.7g, 0.1mol) and triethylamine dropwise at 0°C (14ml, 0.1mol) in anhydrous tetrahydrofuran (50ml) solution, after 2 hours of dropwise addition, slowly rise to room temperature and stir overnight, N 2 Or triethylamine hydrochloride was removed by filtration under an Ar gas atmosphere, most of the solvent tetrahydrofuran was removed under reduced pressure, and the residue was distilled under reduced pressure to collect the product at 120° C. (0.1 mmHg). The product is a colorless oil. Weight 11.3g, yield 75.0%
[0048] in N 2 Or under Ar gas atmosphere, in a 100ml three-necked bottle, add (0.7g, 5.76mmol) and DMAP (140mg, 1.1mmol) were disso...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Melting point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More