Kit for detecting mycobacterium tuberculosis based on blood free nucleic acid
A technology of Mycobacterium tuberculosis and free nucleic acid is applied in the field of test kits for detecting Mycobacterium tuberculosis, which can solve the problems of low diagnosis rate of sputum samples, delayed patient treatment time, low sensitivity, etc., so as to avoid crossover between multiple pathogens. The effect of reaction, avoiding the generation of non-specific amplification products, and reducing the safety risk factor
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0043] The present embodiment provides a kind of test kit (PCR-fluorescence probe method) based on blood free nucleic acid detection Mycobacterium tuberculosis, it comprises PCR reaction solution, primer, enzyme, internal standard, quality control sample, detect MTB target nucleoside Acid fluorescent probes, detection internal standard fluorescent probes, positive quality controls and negative quality controls, primers include detection of MTB target nucleotide primers and detection of internal standard primers, detection of the nucleotide sequence of MTB target nucleotide primers As shown in SEQ ID NO.1, SEQ ID NO.2, SEQ ID NO.3, SEQ ID NO.4 (2 pairs of primers);
[0044] The internal standard is a plasmid whose nucleotide sequence is shown in SEQ ID NO.7, and the concentration of the plasmid is 2×10 4 copies / ml, detect the nucleotide sequence of the internal standard primer as shown in SEQ ID NO.5, SEQ ID NO.6;
[0045] The PCR reaction solution includes the following compo...
Embodiment 2
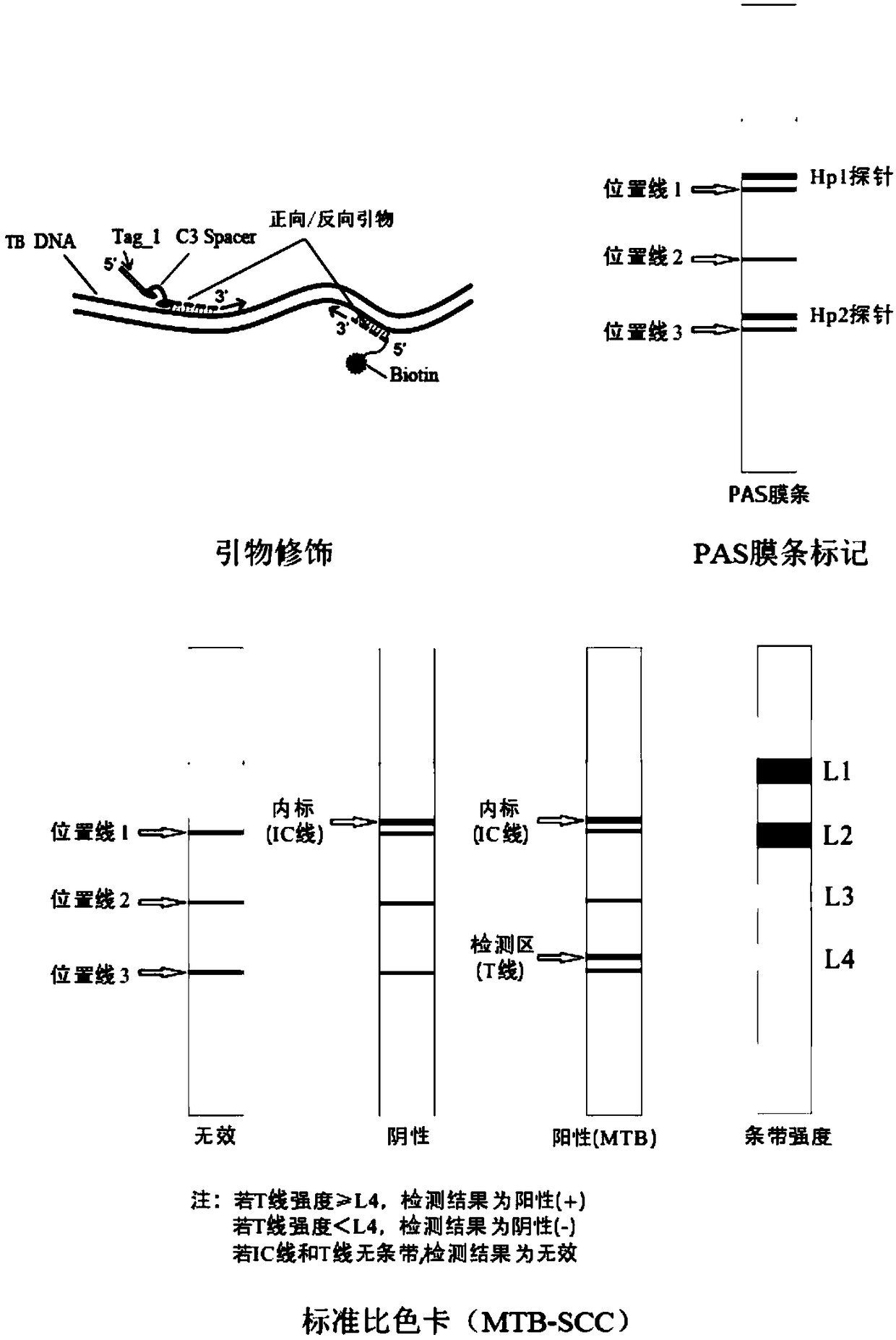
[0083] The present embodiment provides a test kit (PCR-membrane chromatography hybridization method) for detecting Mycobacterium tuberculosis based on blood free nucleic acid, which includes PCR reaction solution, primers, enzymes, internal standard, hybridization membrane strip, chromogenic solution, Positive quality control and negative quality control, with hybridization probes attached to the hybridization membrane strip;
[0084] The internal standard is the plasmid whose nucleotide sequence is shown in SEQ ID NO.7, and the concentration of the plasmid is 2×10 4 copies / ml, the nucleotide sequence of the detection internal standard primer is shown in SEQ ID NO.5, SEQ ID NO.6, and the 5' end of the upstream primer of the detection internal standard primer is also connected with the C3 spacer arm (C3spacer ) and the first tag sequence, the first tag sequence is shown in SEQ ID NO.15; the 5' end of the downstream primer of the detection internal standard primer is also connec...
Embodiment 3
[0123]In this embodiment, the clinical samples are detected by the kits of Examples 1-2 and the sputum smear method. The actual situation of the clinical samples is: 103 cases of pulmonary tuberculosis, 26 cases of extrapulmonary tuberculosis, 43 negative cases, and a total of 172 cases. Two kinds of kits (PCR-fluorescent probe method and PCR-membrane chromatography hybridization method) measurement results of the present embodiment are as follows: figure 2 As shown, and compared with the sputum smear method, the comparison results are as shown in Table 6; Note: Table 6 is the test result of the sputum smear when viewed vertically, and the test results of the two kits of the present invention when viewed horizontally; the views of the following tables similar.
[0124] Table 6
[0125]
[0126]
[0127] As shown in Table 6, the overall positive and negative rates of the sputum smear method were 20.93% (36 / 172) and 79.07% (136 / 172), and the positive rates of pulmonary t...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More