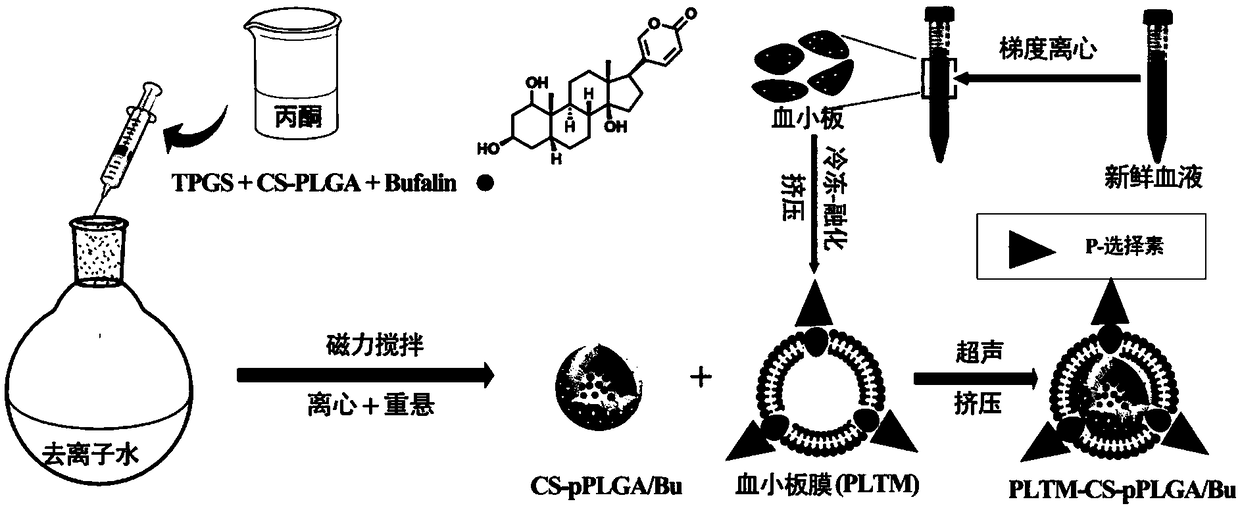
Platelet membrane wrapped drug loading porous nano particle and preparation method thereof
A nanoparticle and platelet technology, applied in the field of drug-loaded nanoparticles and their preparation, can solve the problems of low drug-degrading rate of PLGA nanoparticles, low immune effect and low immunogenicity of PLGA nanoparticles, and achieve excellent results. Biocompatibility, improved biocompatibility, low immunogenicity effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0041] (1) Add 105 mg of PLGA to 10 mL of dimethylformamide solution containing 2.9 mg of EDC·HCl and 1.7 mg of NHS, and activate it in ice bath for 0.5 h. When the temperature was raised to room temperature, 9 mg chitosan oligosaccharide was added and reacted for 48 h under nitrogen protection. Then, it was dialyzed in PBS buffer solution with pH=7.4 for 3 days with a dialysis bag with a molecular weight cutoff of 7000, and then freeze-dried to obtain CS-PLGA.
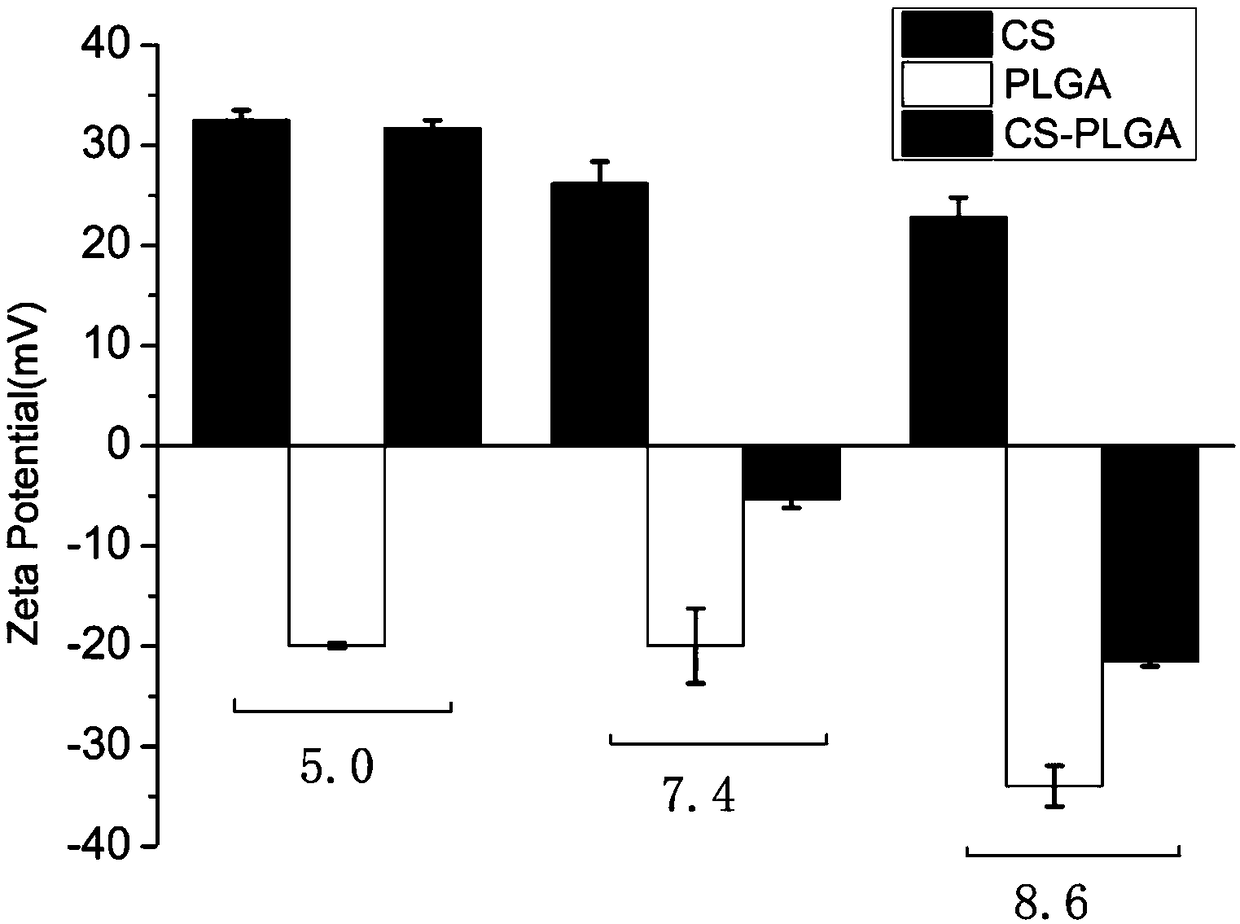
[0042] (2) Disperse the CS-PLGA obtained in step (1), chitosan oligosaccharide, and PLGA in aqueous solutions of pH=5.0, 7.4, and 8.6 to prepare a dispersion of 0.5 mg / mL, and measure its Zeta potential respectively, as figure 2 As shown, when the pH changes from acidic to alkaline, the potential of CS-PLGA changes from positive to negative correspondingly, while the electrical properties of chitosan and PLGA do not change accordingly. The results showed that chitosan oligosaccharides were successfully bonded to PLG...
Embodiment 2
[0044] (1) Dissolve 10mg CS-PLGA and 1mg Bufalin in 1mL acetone, add dropwise to 5mLTPGS 20% (w / v) deionized water at 800rpm, stir in air for 3h, then put in a vacuum oven , to remove residual acetone. Then wash three times with deionized water to remove TPGS, and finally resuspend with the same volume of deionized water to obtain drug-loaded nanoparticles with porous structure. To set up a control, drug-loaded CS-PLGA / Bu nanoparticles without pore structures were prepared in a similar manner to that described above, except that the mixed acetone solution was added to deionized water without TPGS.
[0045] (2) Blood was collected from the heart to obtain fresh blood from female SD rats, and the anticoagulated blood was added to a centrifuge tube containing the separation solution (the volume ratio of the anticoagulation blood to the separation solution was 1:2) and centrifuged at 300 g for 15 minutes. Aspirate the first layer of platelet plasma and add it to another centrifug...
Embodiment 3
[0049] (1) Take 2ml of the CS-PLGA / Bu NPs dispersion, the CS-pPLGA / Bu NPs dispersion and the PLTM-CS-pPLGA NPs dispersion in Example 2 respectively, and pack them into dialysis bags with a molecular weight cut-off of 7000, Then they were respectively immersed in 18 mL of PBS buffer solution with pH=7.4.
[0050] (2) Transfer the above drug release system to a constant temperature shaker at 37°C with a rotation speed of 100rpm for 48h, and take 1mL at 0.25h, 0.5h, 1h, 2h, 4h, 8h, 12h, 24h, and 48h to measure its concentration at 298nm. UV absorbance was measured, and then 1 mL of PBS buffer under the same conditions was re-supplemented, data were collected and drug release was calculated.
[0051] The release profiles of Bufalin in the three granules are as follows Figure 6 shown. After 48h, the cumulative drug release of CS-PLGA / BuNPs without pore structure was less than 20%. However, the cumulative drug release rate of CS-pPLGA / Bu NPs with a porous structure is as high as...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More