Copolyester as well as preparation method and product
A technology of copolyester and products, applied in the field of copolyester and its preparation, can solve the problems of high crystallinity, high melting point, large processing method and application limitations, and achieve the effect of good gas barrier performance and good performance
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0047] The present invention also provides a kind of preparation method of copolyester, described preparation method comprises:
[0048] S1, mixing a dibasic carboxylic acid or its anhydride or its diester with a dibasic alcohol and a catalyst to carry out an esterification reaction to obtain a polyester oligomer;
[0049] S2, mixing the polyester oligomer and the polyglycolic acid oligomer for polycondensation reaction to obtain copolyester.
[0050] In step S1, the molar ratio of the dicarboxylic acid or its anhydride or its diester to the dihydric alcohol is 1:1.1˜1:2.5.
[0051] Considering that the activity of the dibasic carboxylic acid diester is better, it is preferable to carry out the esterification reaction between the dibasic carboxylic acid diester and the dibasic alcohol.
[0052] Specifically, the esterification reaction is carried out under the protection of an inert gas such as nitrogen, and the catalyst used in the esterification reaction is the first catalyst...
Embodiment 1
[0081] The copolyester of this embodiment is defined as PBFGA30, and m:n is 70:30 in PBFGA30, and concrete preparation process is as follows:
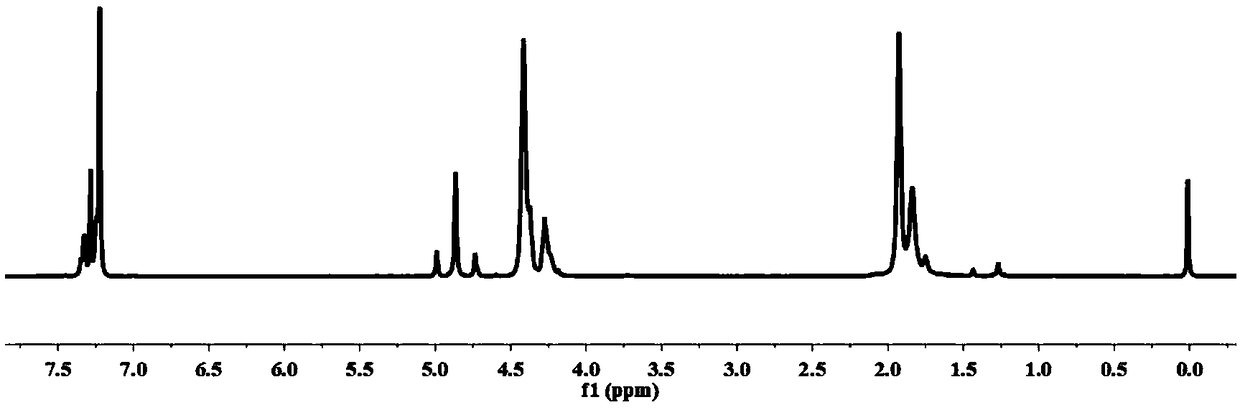
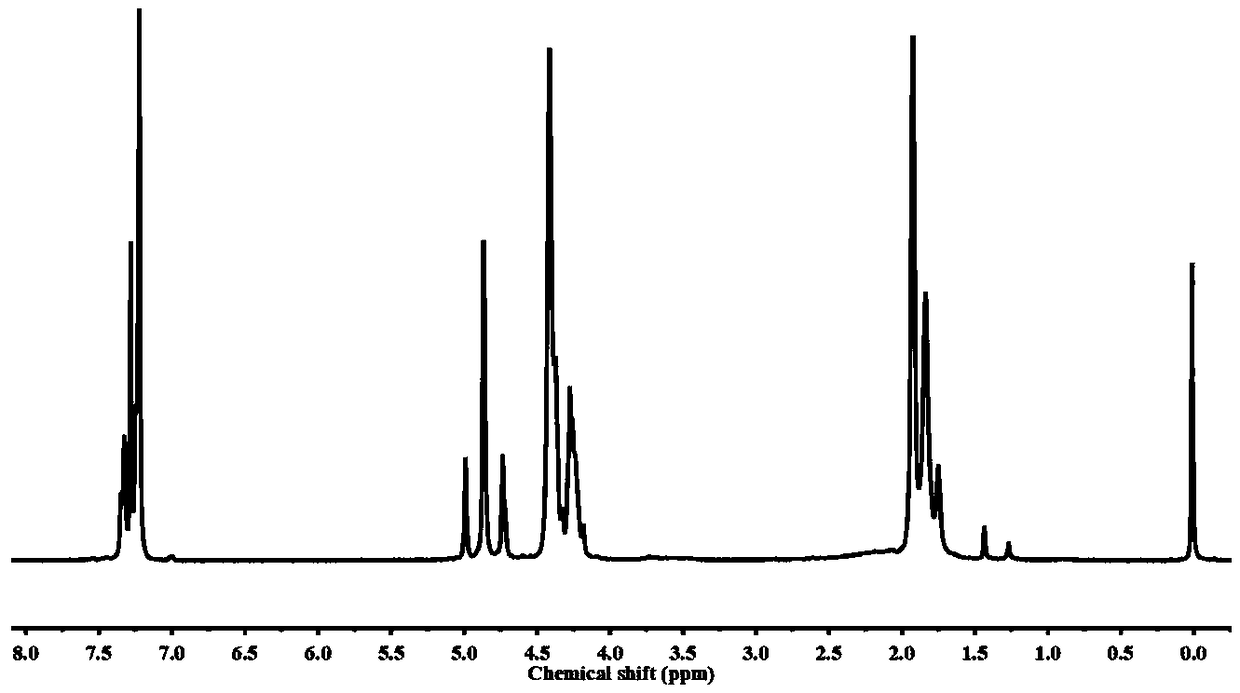
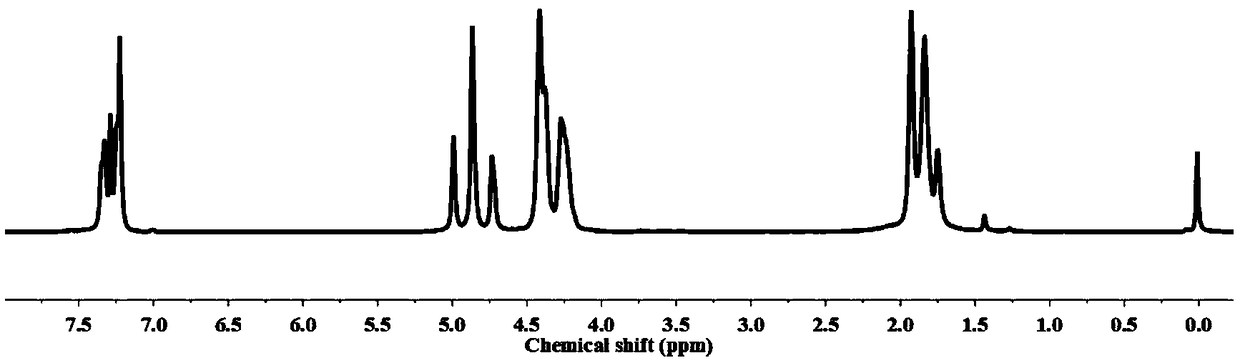
[0082] (1) Preparation of polyester oligomers by esterification: first put 0.5 moles of dimethyl 2,5-furandicarboxylate, 1.25 moles of 1,4-butanediol, and 100 mg of tetrabutyl titanate into the reactor , under the protection of high-purity nitrogen, react at 200° C. for 4 h, until the content of methanol produced exceeds 95% of the theoretical value, and obtain a polyester oligomer with a structural formula such as formula (3).
[0083] (2) Dehydration condensation reaction to prepare polyglycolic acid oligomers: put glycolic acid into the reactor, add zinc acetate, the temperature is 150°C, reduce the pressure to 1000Pa, the dehydration condensation reaction time is 2h, and the obtained structural formula is as follows: ) represented by the polyglycolic acid oligomer, the polyglycolic acid oligomer has a melting point of 145°C.
[00...
Embodiment 2
[0091] The copolyester of this embodiment is defined as PBFGA40, m in PBFGA40: n is 60:40, and concrete preparation process is as follows:
[0092] (1) Preparation of polyester oligomers by esterification: first put 0.5 moles of dimethyl 2,5-furandicarboxylate, 0.55 moles of 1,4-butanediol, and 100 mg of tetrabutyl titanate into the reactor , under the protection of high-purity nitrogen, react at 180° C. for 6 h, until the content of methanol produced exceeds 95% of the theoretical value, and obtain a polyester oligomer with the structural formula shown in formula (3).
[0093] (2) Dehydration condensation reaction to prepare polyglycolic acid oligomers: put glycolic acid into the reactor, add zinc acetate, the temperature is 150 ° C, the pressure is reduced to 300Pa, the dehydration condensation reaction time is 1h, and the structural formula is obtained as formula (4 ) represented by the polyglycolic acid oligomer, the polyglycolic acid oligomer has a melting point of 140°C....
PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
| melting point | aaaaa | aaaaa |
| melting point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More