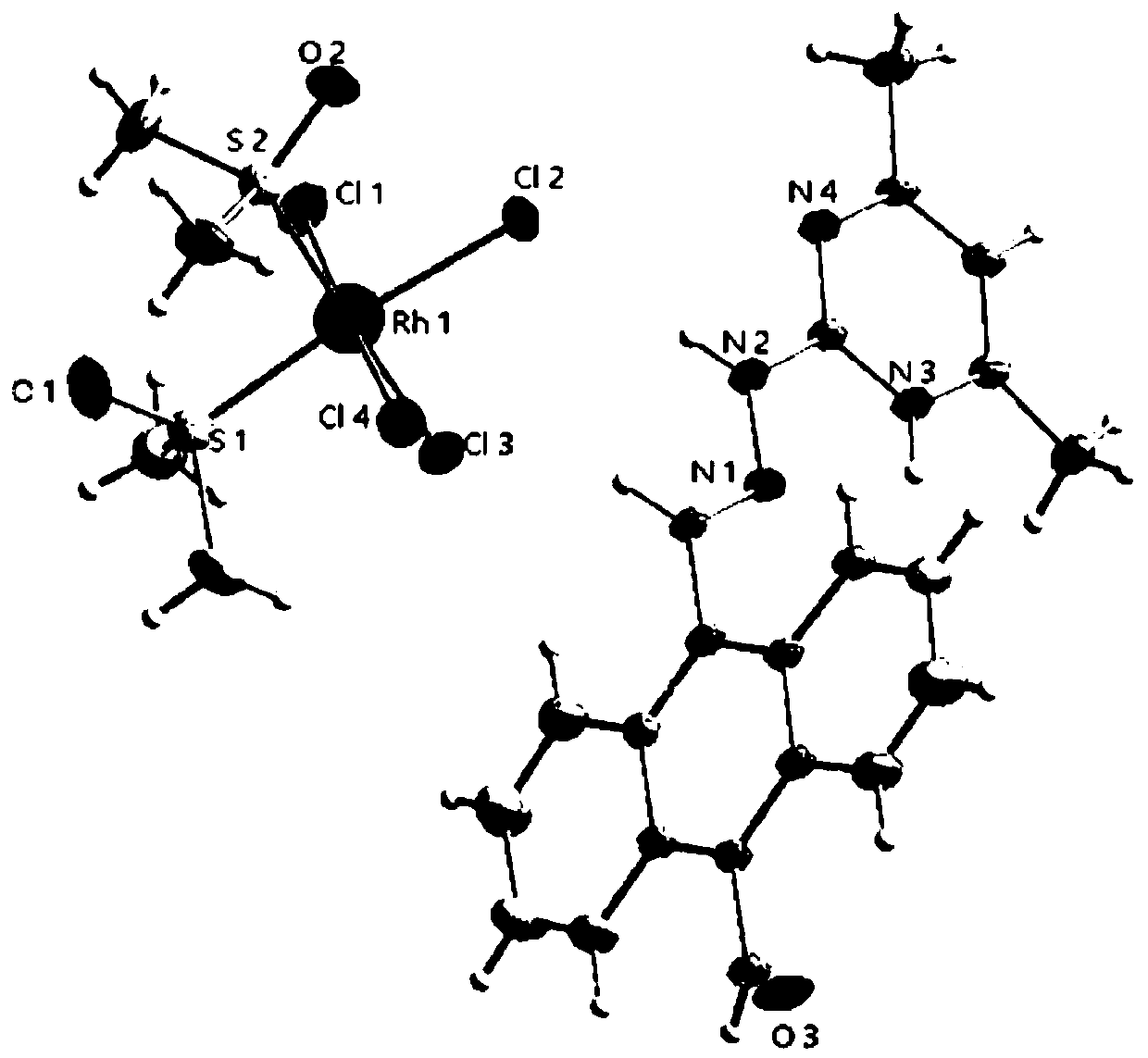
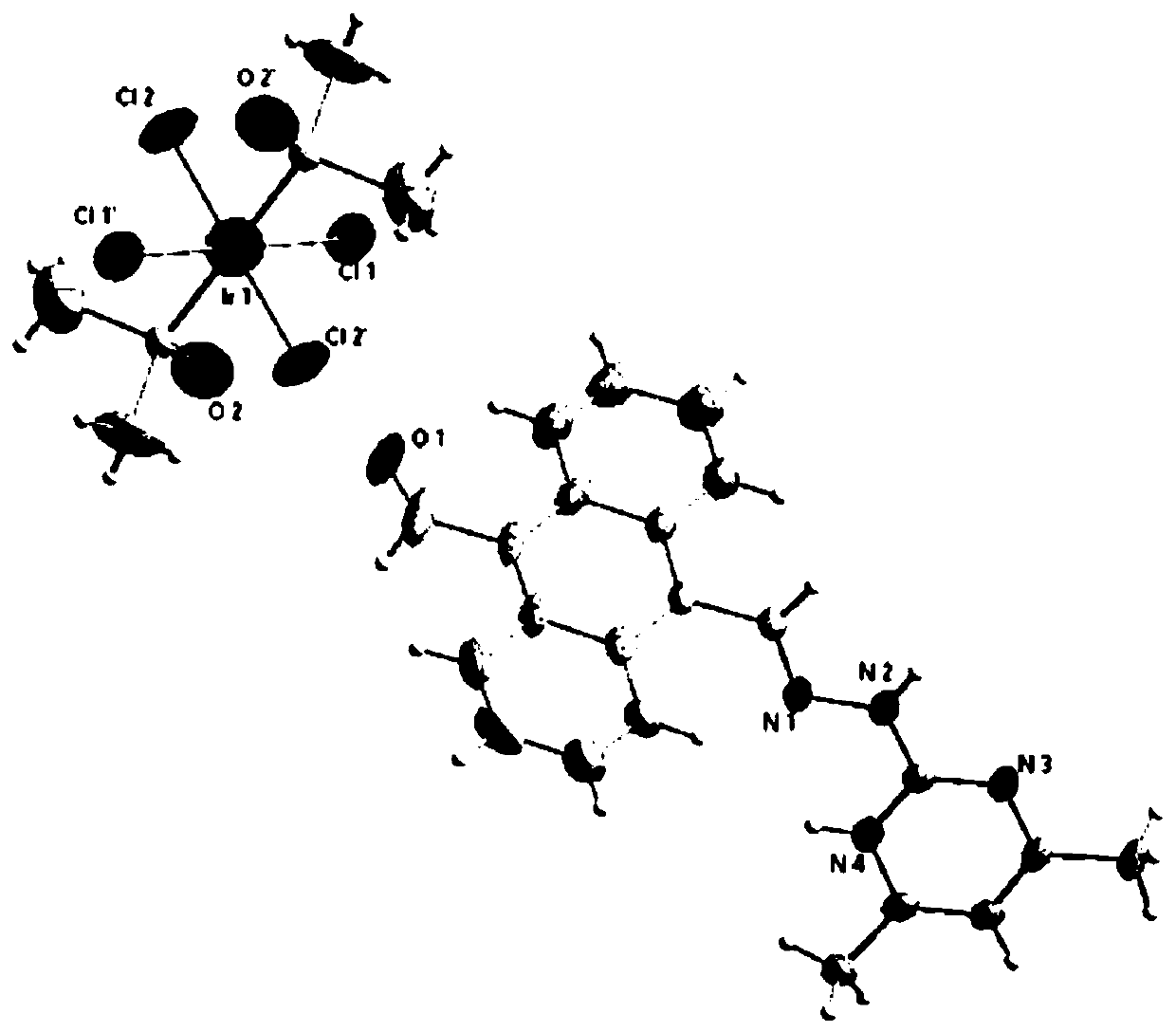
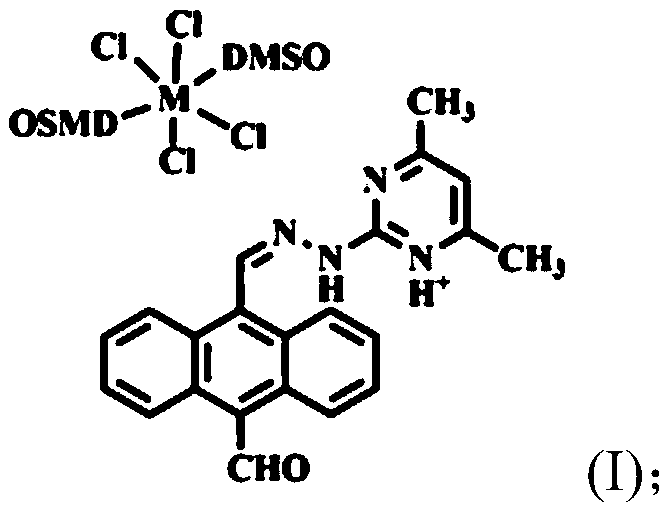
Ionic metal coordination compounds taking 9-aldehyde-10-pyrimidine anthracene hydrazone as ligand, synthesis method and applications thereof
A synthesis method and compound technology, applied in the field of medicine, can solve problems such as difficulty in effectively reducing liver and kidney toxicity, and achieve the effects of significant proliferation inhibitory activity, high activity, good biological affinity and compatibility
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0024] Embodiment 1: Preparation of the compound shown in formula (II) (i.e. 9-formyl-10-(4',6'-dimethylpyrimidine) anthrahydrazone, hereinafter also referred to as ligand)
[0025] 1) Dissolve 5.0mmol of 9,10-anthracenedicarbaldehyde and 6.5mmol of 2-hydrazino-4,6-dimethylpyrimidine in 100mL of methanol, reflux at 65°C, and track and detect the reaction with TLC Complete (about 8h), stop the reaction, filter the reaction solution while it is hot, and dry the obtained solid in vacuum for 10h to obtain a mixed product with a yield of 85%;
[0026] 2) The resulting mixed product is eluted by silica gel column chromatography with a mixed eluent composed of petroleum ether and dichloromethane (volume ratio of petroleum ether and dichloromethane is 1:3), while tracking with thin layer chromatography Monitor, collect the eluate containing the target product, concentrate and dry it to obtain a yellow solid product with a yield of 65%.
[0027] Infrared spectrum, elemental analysis, ...
Embodiment 2
[0035]Embodiment 2: the preparation of compound shown in formula (II)
[0036] Example 1 was repeated, except that in step 2), a mixed eluent composed of petroleum ether and dichloromethane in a volume ratio of 1:1 was used for elution.
[0037] As a result, a yellow solid product was obtained with a yield of 30%.
[0038] Infrared spectrum, electrospray mass spectrometry, elemental analysis, 1 H NMR spectrum and 13 C nuclear magnetic resonance spectrum identification, determined to be the compound shown in formula (II).
Embodiment 3
[0039] Embodiment 3: the preparation of compound shown in formula (II)
[0040] Repeat Example 1, the difference is:
[0041] In step 1), ethanol is replaced with methanol, and the reaction is carried out at 80° C.;
[0042] In step 2), a mixed eluent composed of petroleum ether and dichloromethane at a volume ratio of 1:5 was used for elution.
[0043] As a result, a yellow solid product was obtained with a yield of 45%.
[0044] Infrared spectrum, electrospray mass spectrometry, elemental analysis, 1 H NMR spectrum and 13 C nuclear magnetic resonance spectrum identification, determined to be the compound shown in formula (II).
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More