Synthesis method for co-production of N-hydroxyethyl piperazine and N-ethyl piperazine
A technology of hydroxyethylpiperazine and ethylpiperazine, applied in the field of synthesis of N-hydroxyethylpiperazine co-production of N-ethylpiperazine, can solve the problem of high raw material prices affecting the production of N-ethylpiperazine Efficiency, danger and other issues, to achieve the effect of low raw material price, good industrialization prospect and high conversion rate
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
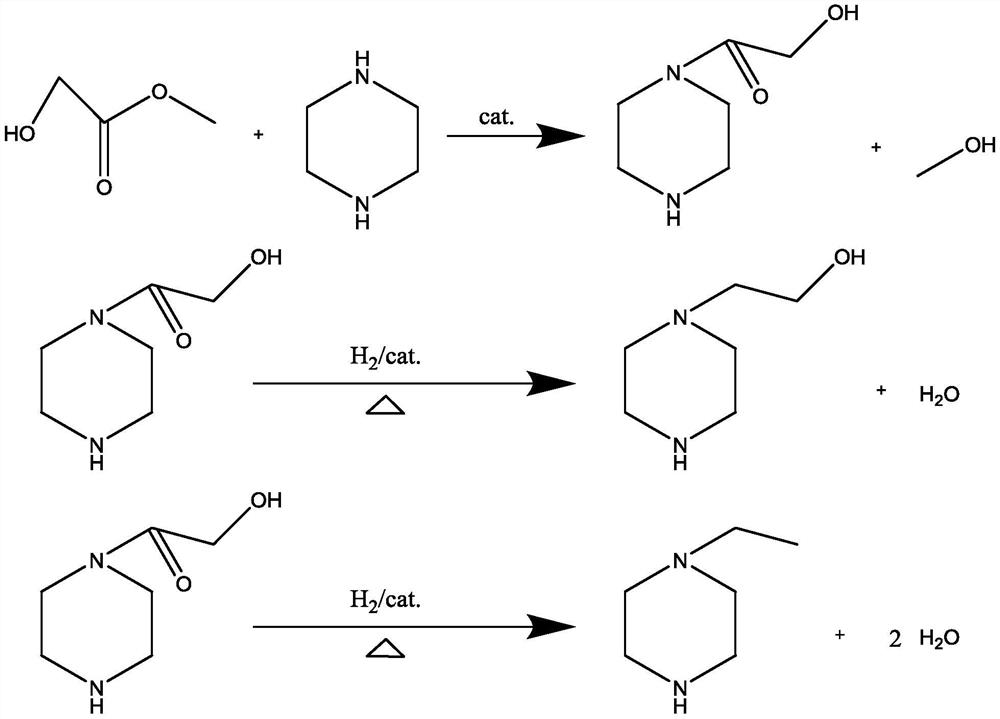
[0030] Take 172g (2mol) of anhydrous piperazine into a four-neck flask with mechanical stirring, add 344g of anhydrous methanol to dissolve, add 5.4g (0.1mol) of anhydrous sodium methoxide, stir at 600-700rpm, and slowly drop in Add 172.8g (1.92mol) methyl glycolate, control the dropping time for 6 hours, and control the temperature during the dropping process to 30°C; after the dropping is completed, stir at 300-500rpm for 2 hours, turn on the vacuum system, recover the solvent, and take samples in the middle to detect until The solvent is recovered cleanly, and the material is transferred into an autoclave;
[0031] Add 344g of toluene, put in 17.2g of Raney nickel as a catalyst, tighten the lid of the kettle, replace the air three times after the nitrogen leak test, and replace it with hydrogen twice;
[0032] Turn on the stirring at 400-600rpm, raise the temperature at the same time, raise the temperature to 200°C, fill in hydrogen to keep the pressure at 5±0.5MPa, observe...
Embodiment 2
[0035] This implementation method is different from Example 1 in that the ammonium solution catalyst is 10.6g (0.1mol) of sodium carbonate, and the other steps are the same to obtain N-ethylpiperazine 59.51g with a purity of 99.5%; N-hydroxyethylpiperazine 158.34 g, purity 99.7%, total molar yield 87.0%.
Embodiment 3
[0037] The difference between this implementation method and Example 1 is that the ammonium solution catalyst is 10.0 g (0.1 mol) of potassium bicarbonate, and the other steps are the same to obtain 57.68 g of N-ethylpiperazine with a purity of 99.6%; N-hydroxyethylpiperazine 152.36g, purity 99.6%, total molar yield 83.9%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More