Modified binding molecules comprising connecting peptides
a technology of connecting peptides and binding molecules, which is applied in the direction of antibody mimetics/scaffolds, fusion polypeptides, dna/rna fragmentation, etc., can solve the problems of increasing the effort required for downstream processing, affecting the overall yield, and difficult separation of form a and form b, so as to enhance the formation of form a
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Identification of A and B Isoforms
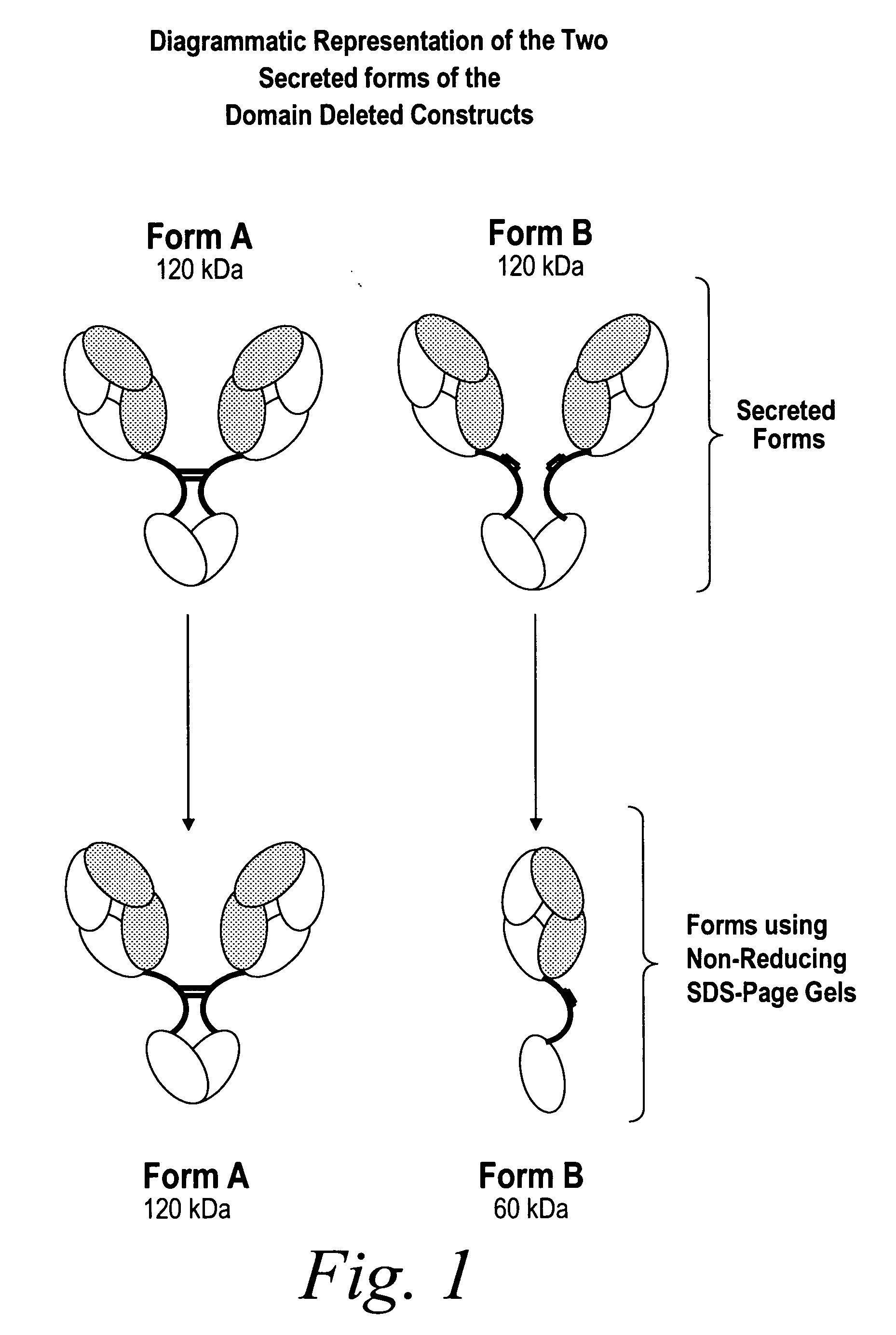
[0403] Solutions of antibody molecules comprise two different isoforms. One form, Form A comprises heavy chain molecules that are linked via at least one disulfide linkage. The other form, Form B, comprises heavy chain molecules that are not linked via at least one disulfide linkage. Form B does not appear or appears at a very low frequency in with intact gamma 1 MAbs, such as Rituxan®. However with domain deleted (dd) constructs having a similar hinge, the frequency of Form B is much higher. These forms can be distinguished using denaturing, non-reducing SDS page. In domain deleted antibody preparations, Form A appears as a 120 kDa dimer while form B appears as a 60 kDa monomer (FIG. 1).
example 2
Identification of Hinge Region Heterogeneity in CH2 Domain Deleted MAb Fragments
[0404] Hinge domains can be subdivided into three distinct regions: upper, middle, and lower hinge regions (Roux et al. J. Immunol. 1998 161:4083). Polypeptide sequences encompassing these regions for IgG1 and IgG3 hinges are shown in Table 1. The IgG3 hinge middle region contains, in addition to the two conserved cysteine residues, a 15 amino acid motif that repeats three times. Amino acid sequences from these regions were used to design synthetic IgG1 / IgG3 connecting peptides. These consisted of IgG1 upper hinge residues corresponding to positions 226 through 238, an IgG1 middle hinge corresponding to positions 239 through 241, and a single IgG3 middle hinge repeat motif corresponding to positions 241 EE through 242 combined with either an added proline at position 243 or an added proline, alanine, proline at positions 243, 244, and 245, respectively (Kabat numbering system), followed by a flexible Gl...
example 3
Construction of Connecting Polypeptides and Preferential Synthesis of Isoforms
[0406] Nucleic acid sequences encoding the hinge region connecting peptides shown in Table 2 were introduced into CH2 domain deleted huCC49 gene sequences using the Splicing by Overlap Extension (SOE) method (Horton, R. M. 1993 Methods in Molecular Biology, Vol 15: PCR Protocols: Current Methods and applications. Ed. B. A. White). Correct modifications to the hinge region were confirmed by DNA sequence analysis. Plasmid DNA was used to transform CHO DG44 cells for stable production of antibody protein.
[0407] CH2 domain deleted huCC49 antibodies containing the eight designed synthetic connecting peptides indicated in Table 2 were constructed and antibody produced in CHO DG44 cells. Supernatants were collected from isolated cell lines and concentration of antibody in the culture supernatants determined by immunoassay. Supernatants containing antibody ranging from 0 to 30 ng of total antibody protein from e...
PUM
| Property | Measurement | Unit |
|---|---|---|
| MW | aaaaa | aaaaa |
| MW | aaaaa | aaaaa |
| molecular mass | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More