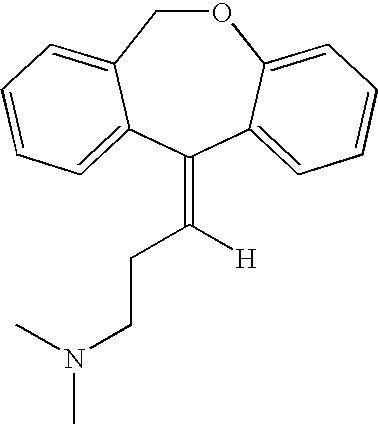
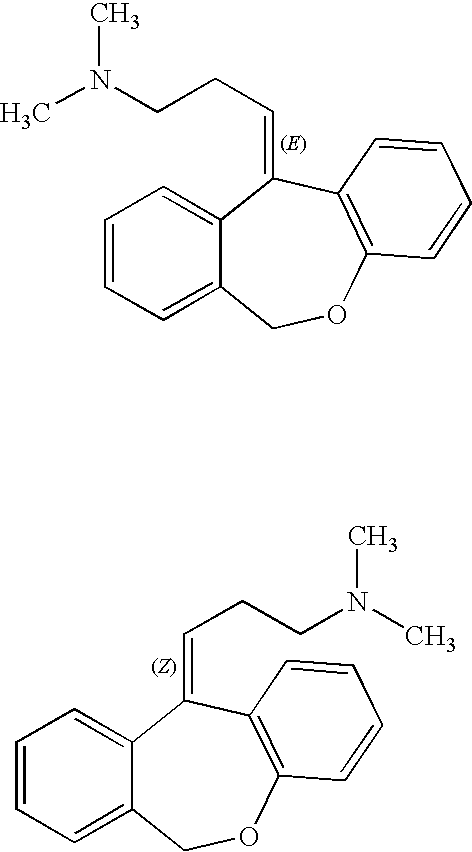
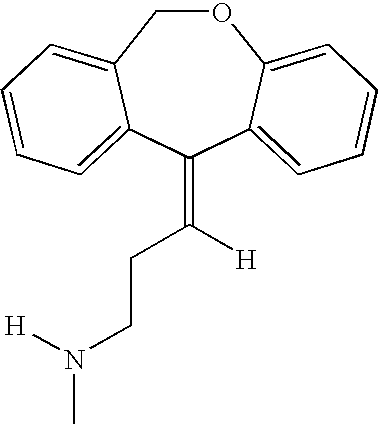
Doxepin isomers and isomeric mixtures and methods of using the same to treat sleep disorders
a technology of isomeric mixtures and doxepins, which is applied in the direction of biocide, drug compositions, nervous disorders, etc., can solve the problems of many physicians being reluctant to prescribe benzodiazepines, withdrawal symptoms such as agitation, rebound insomnia, confusion,
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0112]Doxepin is prepared by the following method.
[0113](a) A Grignard compound is prepared in the conventional manner from 4.8 g (0.2 gram-atom) magnesium in 100 ml ether and 30 g (34 ml) (3-chloropropyl)-tertbutyl-ether and 16.40 grams (0.078 mol) 6,11-dihydrodibenzo-[b,e]-oxepine-11-one dissolved in 100 ml ether is added in dropwise fashion so that the contents of the flask boil lightly. The mixture is heated for 1 hour with agitation in a reflux condenser to complete the reaction and then it is decomposed with ammonium chloride solution. The product which is obtained by separating, drying and eliminating the solvent produced, when the ether residue (24.0 g) is extracted with ligroin, amounts to 20.3 g (80.0% of theory) of 11-(3-tertbutoxypropyl)-11-hydroxy-6,11-dihydrodibenzo-[b,e]-oxepine, having a melting point of 124-126° C. The (3-chloropropyl)-tertbutyl ether is thereafter obtained in the following manner: 19 g (0.2 mol) 1-chloropropanol-(3), 50 ml liquid isobutylene and 0....
example 2
Preparation of Doxepin Isomer
[0117]Five grams of the cis / trans mixture of doxepin hydrochloride is converted to the free base and then to the maleate salt, M.P. 168-169° C. Several recrystallizations from ethanol afford a pure isomeric maleate, M.P. 172-173° C. This is reconverted to the free base and then to the hydrochloric acid-addition salt. This is purified by recrystallization from a mixture of ethanol and ether; M.P. 192-193° C. The other pure isomer is isolated by the concentration of the crystallization liquors to dryness, followed by converting the residue to the free base and then converting the base to the hydrochloric-acid addition salt. After re-crystallization from a mixture of ethanol and ether, the salt has a M.P. of 209-210.5° C.
example 3
Preparation of Desmethyldoxepin
[0118]Desmethyldoxepin is prepared according to the following method. Anhydrous 3-methylaminopropyltriphenylphosphonium bromide hydrobromide (1530 g) prepared as in U.S. Pat. No. 3,509,175, is suspended in 4.5 l dry tetrahydrofuran and 6.0 moles of butyl lithium in heptane is added during 1 hour. After an additional 30 minutes, 483 g of 6,11-dihydrodibenz(b,e)oxepin-11-one, is added to the deep red solution and the reaction is maintained at reflux for 10 hours. Water, 500 ml, is added at room temperature and the solvent is removed in vacuo. The crude residue is treated with 10% hydrochloric acid until acidic (pH 2) and then 1.5 l benzene is added. After stirring, the mixture separates into three phases (an insoluble hydrochloride salt product phase, an aqueous phase and an organic phase). The benzene layer is removed by decantation and the remaining mixture is rendered basic with 10% sodium hydroxide solution and is extracted with 3×1500 ml portions of...
PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
| boiling point | aaaaa | aaaaa |
| melting point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More