Methods and compositions for oral administration of exenatide
a technology of exenatide and composition, which is applied in the direction of drug composition, peptide/protein ingredient, metabolic disorder, etc., can solve the problems of low patient compliance of parenterally administered peptide drugs, relative ineffectiveness of peptide drugs given perorally, and blood sugar to become too low, so as to reduce food intake, reduce food intake in a subject, and reduce gastric motility
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Protection of Exenatide from Proteases and Successful Administration Via the Duodenum in Dogs
Materials and Experimental methods
Formulations
[0095](1) A formulation containing 150 milligram (mg) Na-EDTA (Sigma-Aldrich, St. Louis, Mo.), 125 mg soybean trypsin inhibitor (SBTI; Sigma), 50 μg exenatide, and 0.8 milliliter (ml) fish oil was prepared.
[0096]A formulation containing 150 milligram (mg) Na-EDTA (Sigma-Aldrich, St. Louis, Mo.), 125 mg soybean trypsin inhibitor (SBTI; Sigma), 100 μg exenatide, and 0.8 milliliter (ml) fish oil was prepared.
[0097](3) A formulation containing 150 milligram (mg) Na-EDTA (Sigma-Aldrich, St. Louis, Mo.), 125 mg soybean trypsin inhibitor (SBTI; Sigma), 0.8 milliliter (ml) fish oil was prepared.
Results
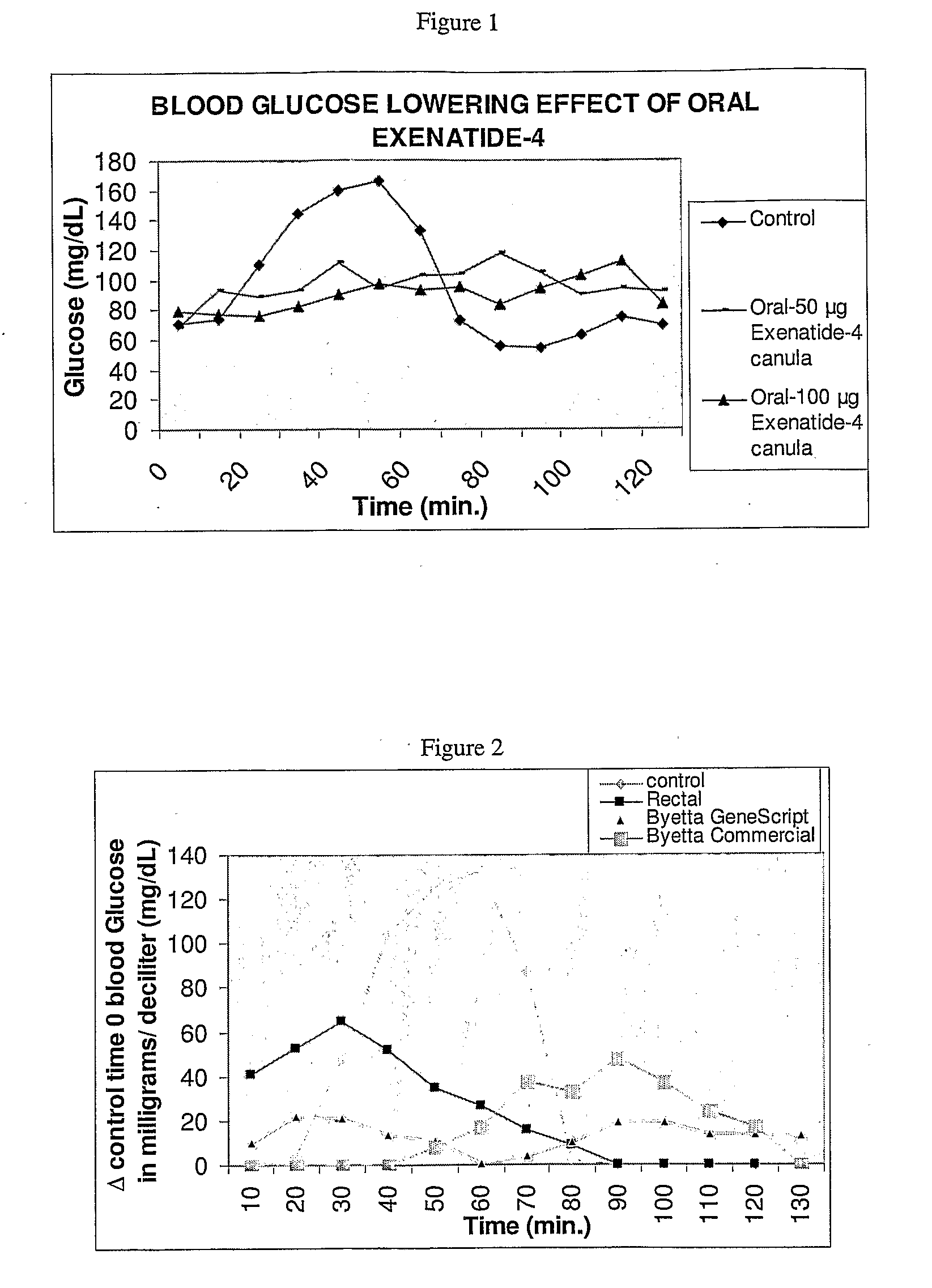
[0098]To test whether exenatide can be protected from proteases and absorbed via the duodenum, formulation 1 (treatment) or 3 (control) were administered directly to the duodenum of about 9 kg beagle dogs or by an endoscope to about 16 kg pigs.
[0099]All dog...
example 2
Injectable Dosage Forms Compared to Rectal and Oral Dosage Forms
Materials and Experimental Methods
Formulation
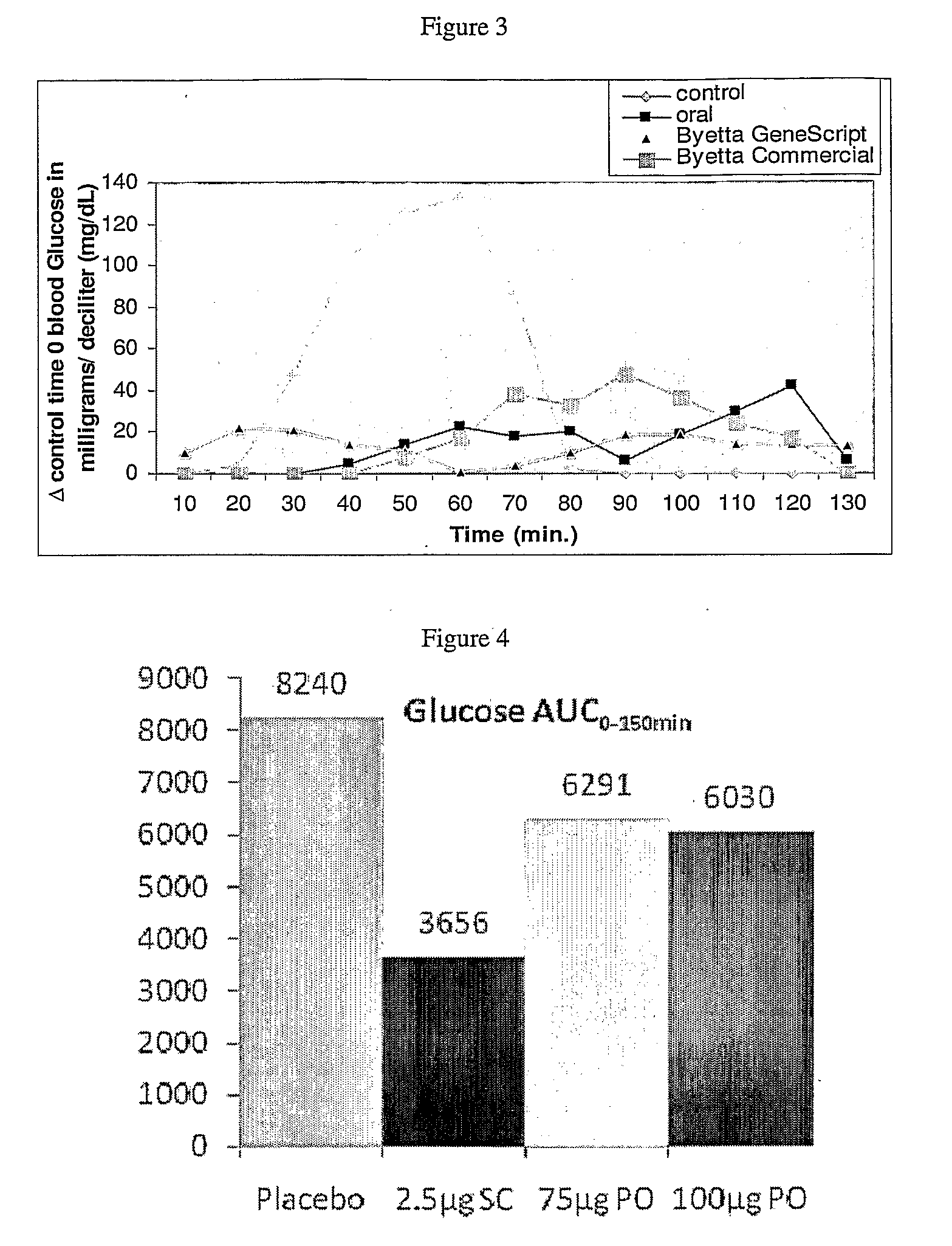
[0102]The following formulations were prepared:[0103](1) An injectable formulation comprising 2.5 μg commercial byetta.[0104](2) An injectable formulation comprising 2.5 μg GeneScript (Piscataway, N.J.) byetta.[0105](3) An oral formulation containing 150 milligram (mg) Na-EDTA (Sigma-Aldrich, St. Louis, Mo.), 125 mg soybean trypsin inhibitor (SBTI; Sigma), 50 μg exenatide (GeneScript (Piscataway, N.J.)) and 0.8 milliliter (nil) fish oil.[0106](4) An oral formulation containing 150 milligram (mg) Na-EDTA (Sigma-Aldrich, St. Louis, Mo.), 125 mg soybean trypsin inhibitor (SBTI; Sigma), 0.8 milliliter (ml) fish oil.[0107](5) A hard gelatin capsule (rectal) containing 150 milligram (mg) Na-EDTA (Sigma-Aldrich, St. Louis, Mo.), 125 mg soybean trypsin inhibitor (SBTI; Sigma), 50 μg exenatide (GeneScript (Piscataway, N.J.)) and in 0.8 milliliter (ml) fish oil.
Results
[0108]To test the...
example 3
Enteral Administration of Exenatide-4; Proof of Concept Pharmacodynamic Study in Dogs with a Formulation Which Facilitates the Absorption of Exenatide-4 Across Biological Membranes
Materials and Experimental Methods
[0113]Study was conducted in 4 beagle dogs with an average weight of 10 kg. All the dogs had a cannula residing in the jejunum through which the drug was administered. After an overnight fast, the dogs were given different doses of oral GLP-1 analogue or sc injection of the analogue. Absorption of the GLP-1 analogue was assessed by measuring the effect on glucose excursion following an oral glucose load. Control experiment consisted of oral dosing without administration of GLP-1 analogue. The interval between oral administration and the oral glucose load was 30 minutes. The primary efficacy end point was the glucose excursion above the pre-OGTT glucose level over a 150 min interval (incremental area under the curve (AUC)0-150 min).
Results
[0114]Direct jejunal instillation o...
PUM
| Property | Measurement | Unit |
|---|---|---|
| weight | aaaaa | aaaaa |
| resistance | aaaaa | aaaaa |
| weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More