[0003]Making fuels and chemicals from biomass is complicated by the need to separate and to purify the intermediate platform molecules at high yields. Conventional approaches to making useful chemicals from biomass typically require very difficult and economically unfeasible separation and purification steps. Disclosed herein is a method in which
levulinic acid (LA) is first produced from biomass (e.g.,
corn stover) by cellulose deconstruction. The LA so formed is then extracted from the aqueous phase using one or more
alkylphenol (AP) solvents. The LA so formed is a value-added, platform compound that finds commercial use as an intermediate or reactant in a host of industrially useful processes. Additionally, the LA may optionally be hydrogenated to γ-
valerolactone (GVL). This strategy allows for any acid that may be used in the process (preferably
sulfuric acid) to be recycled very nearly completely (>99%).
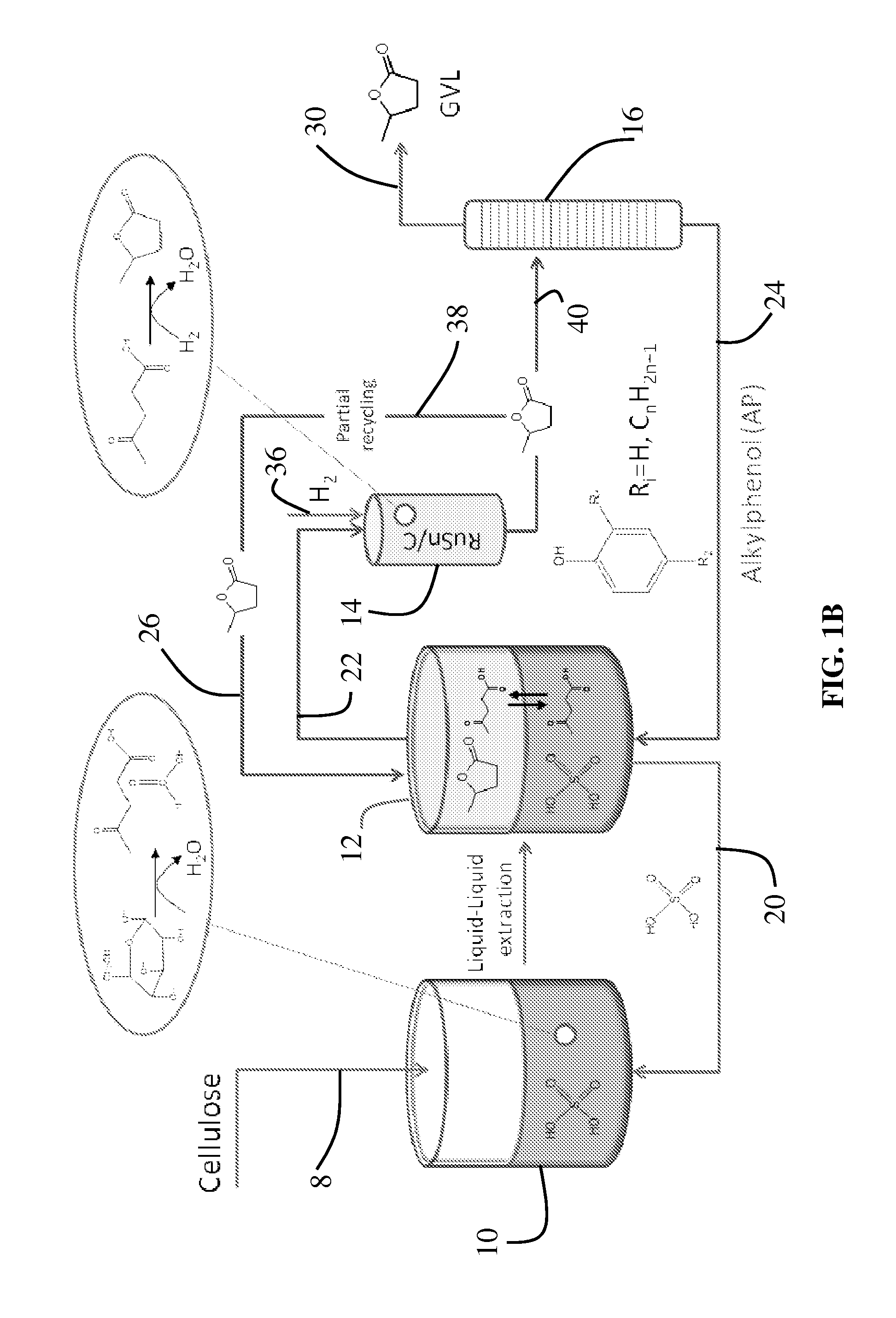
[0008]A distinct
advantage of the method is that the GVL product is stable during the hydrogenation of LA. Thus, it is possible to increase the GVL concentration in the
product mix by successive cycles of cellulose deconstruction, LA extraction, and LA hydrogenation to GVL. By accumulating a large concentration of GVL in the
product mix, GVL can be easily (and cost-effectively) separated from the AP
solvent by simple
distillation.
[0010]Another
advantage of this strategy is that the lower degrees of functionality and boiling points of these platform molecules allow for catalytic
processing in the
vapor phase and / or in organic solvents. This alleviates the need to develop heterogeneous catalysts that are stable under more demanding hydro-
thermal reaction conditions. One such platform molecule is
levulinic acid (LA) (3, 4) from which a variety of fuels and chemicals can be made, such as
valeric acid esters (5), methyltetrahydrofuran (6-8), and esters and ketals of LA (9). Another building block from the reduction of LA is γ-
valerolactone (GVL) (10, 11), which can be used directly as a fuel additive (12), or as a precursor for fuels (13) and chemicals (14, 15). While LA can be formed in significant yields (>50%) by cellulose deconstruction in aqueous solutions of mineral acids such as
sulfuric acid (SA) (16, 17), a challenge for profitable, large-scale production of LA and its derivatives has been separating the LA from the
mineral acid used in the process. This is necessary so that the LA can be further processed downstream without the negative effects of the
mineral acid (18).
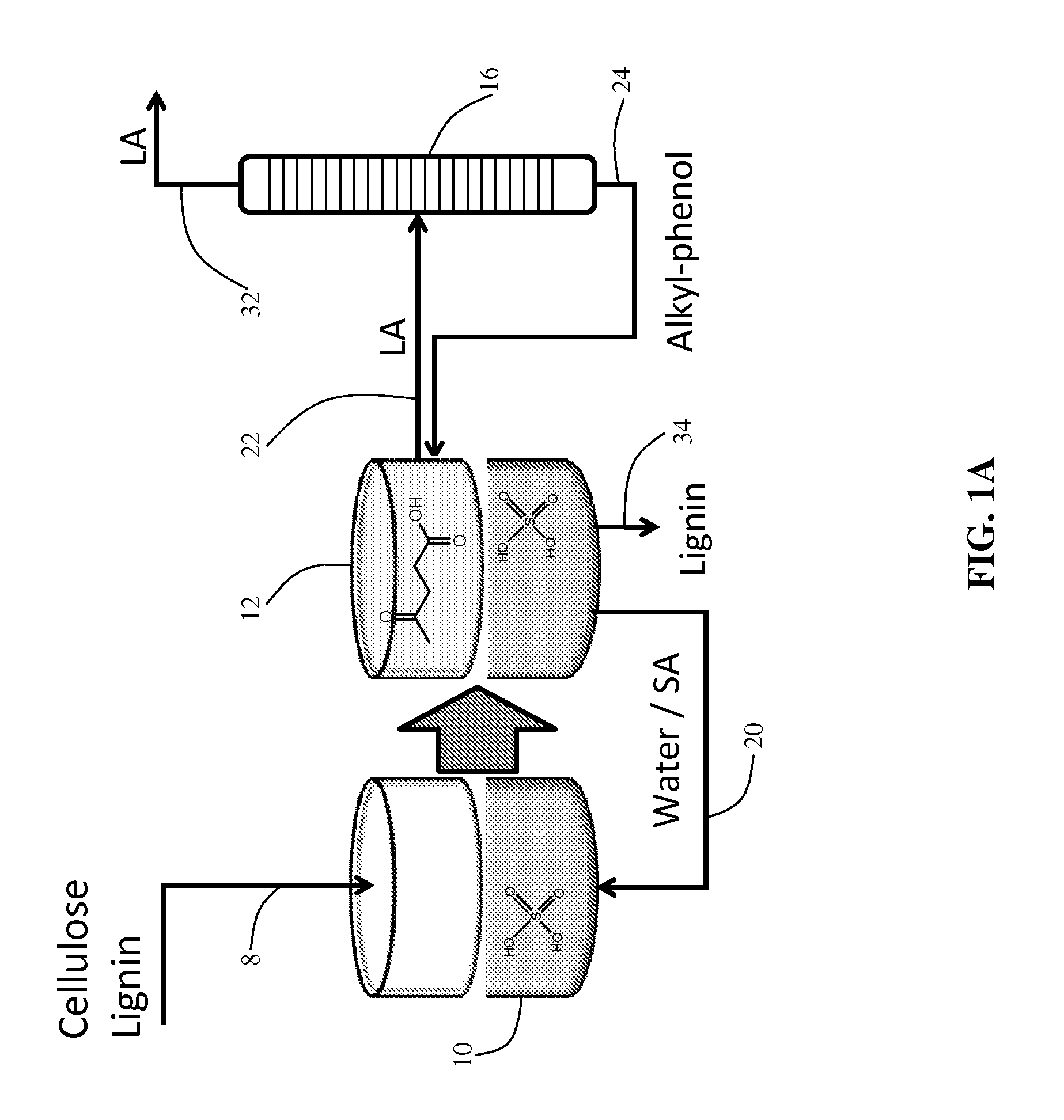
[0011]In the
present method,
alkylphenol (AP) solvents (19) are used as a partitioning agent in a method to produce LA and / or GVL from biomass in general, and
lignocellulosic biomass in particular. As illustrated schematically in FIG. 1, AP solvents can be used to selectively extract LA from aqueous solutions after a cellulose deconstruction step. In addition, AP solvents extract GVL from water with a higher
partition coefficient (the concentration of the solute in the organic phase divided by the concentration of the solute in the aqueous phase) compared to LA. Accordingly, the GVL concentration in the AP
solvent can be increased by the conversion of LA to GVL, combined with the recycle of this
stream for successive extractions. Importantly, a preferred carbon-supported RuSn catalyst (20, 21) can be used to selectively reduce LA to GVL by hydrogenation in the presence of AP, without hydrogenation of the
solvent. This is a critical discovery in that it enables the aforementioned recycling strategy for enhancing the GVL concentration. This then enables the easy and cost-efficient
recovery of the GVL from the AP solvent by simple
distillation. After distillation of GVL from the organic phase, the aqueous phase containing any residual LA and possibly acid (after extraction with the AP solvent) can be recycled for subsequent cycles of cellulose deconstruction, providing an effective strategy for managing the acid used in the deconstruction of the incoming biomass.
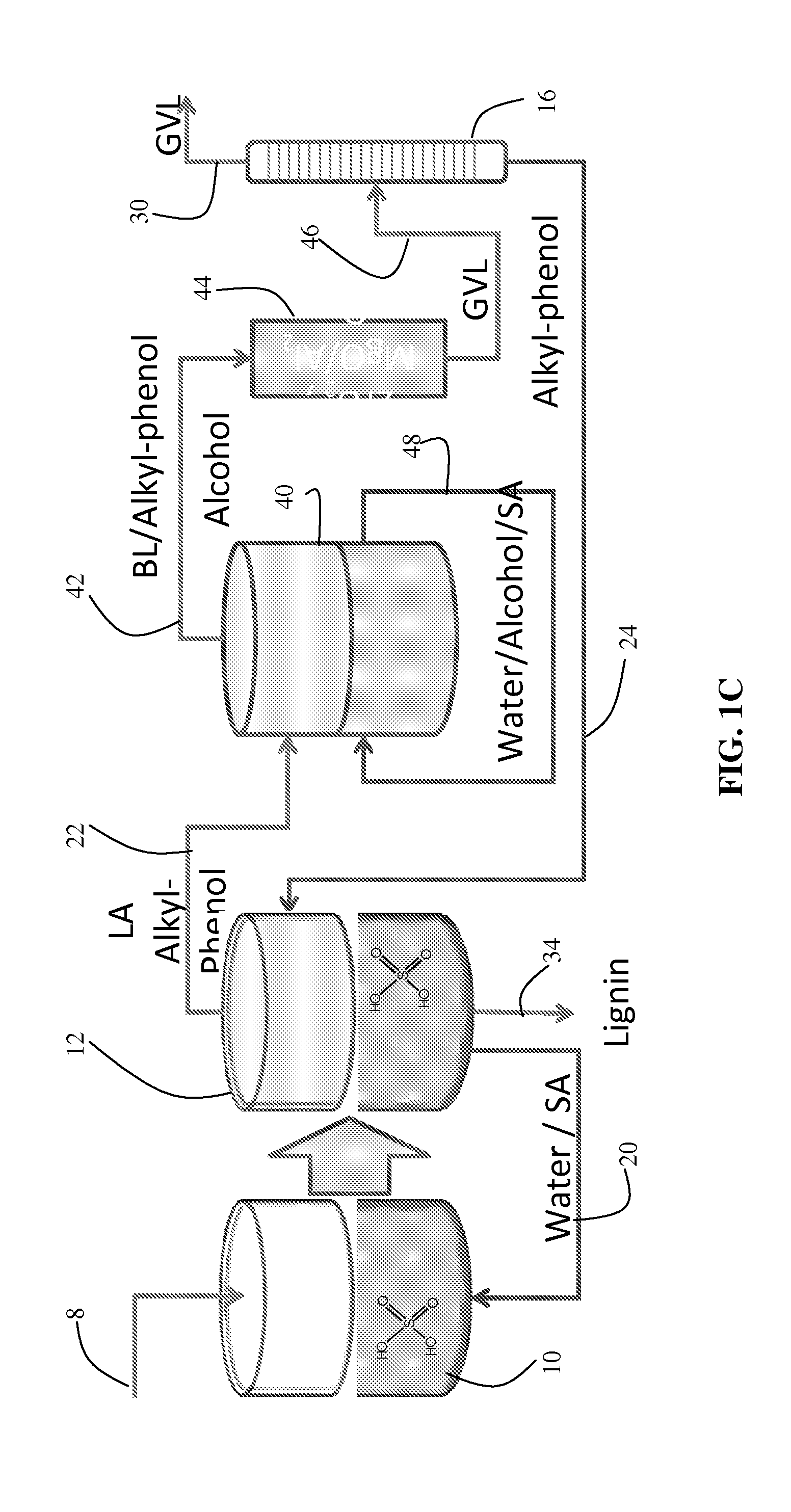
[0013]APs do not partition water or mineral acids. Thus, if an aqueous acid solution is used to deconstruct the incoming cellulose, it may be recovered entirely and recycled accordingly. The relatively high
boiling point of APs allows LA and / or GVL to be distilled from the organic phase without vaporizing water. The APs are organic solvents that are
inert under many upgrading reactions, such as selective hydrogenation. Because the APs have very high partition coefficients for GVL, the GVL can remain in the organic phase when LA is extracted from an
aqueous solution. This allows the GVL concentration in the product
stream to be increased by recycling the product
stream for additional cycles of LA extraction from the aqueous phase containing LA. GVL also increases the extraction of
formic acid from the
aqueous reaction solution.
[0014]Another
advantage is that the ultimate products (either LA or GVL) can be recovered by distillation without having to evaporate water—only the product itself is evaporated. The AP solvent does not require any further
evaporation or separation, and can be directly recycled for additional extractions. Likewise the
aqueous reaction solution used to deconstruct the incoming biomass can also be recycled without further purification. This solution may contain homogeneous acid catalysts, which are recovered intact after extraction.
 Login to View More
Login to View More