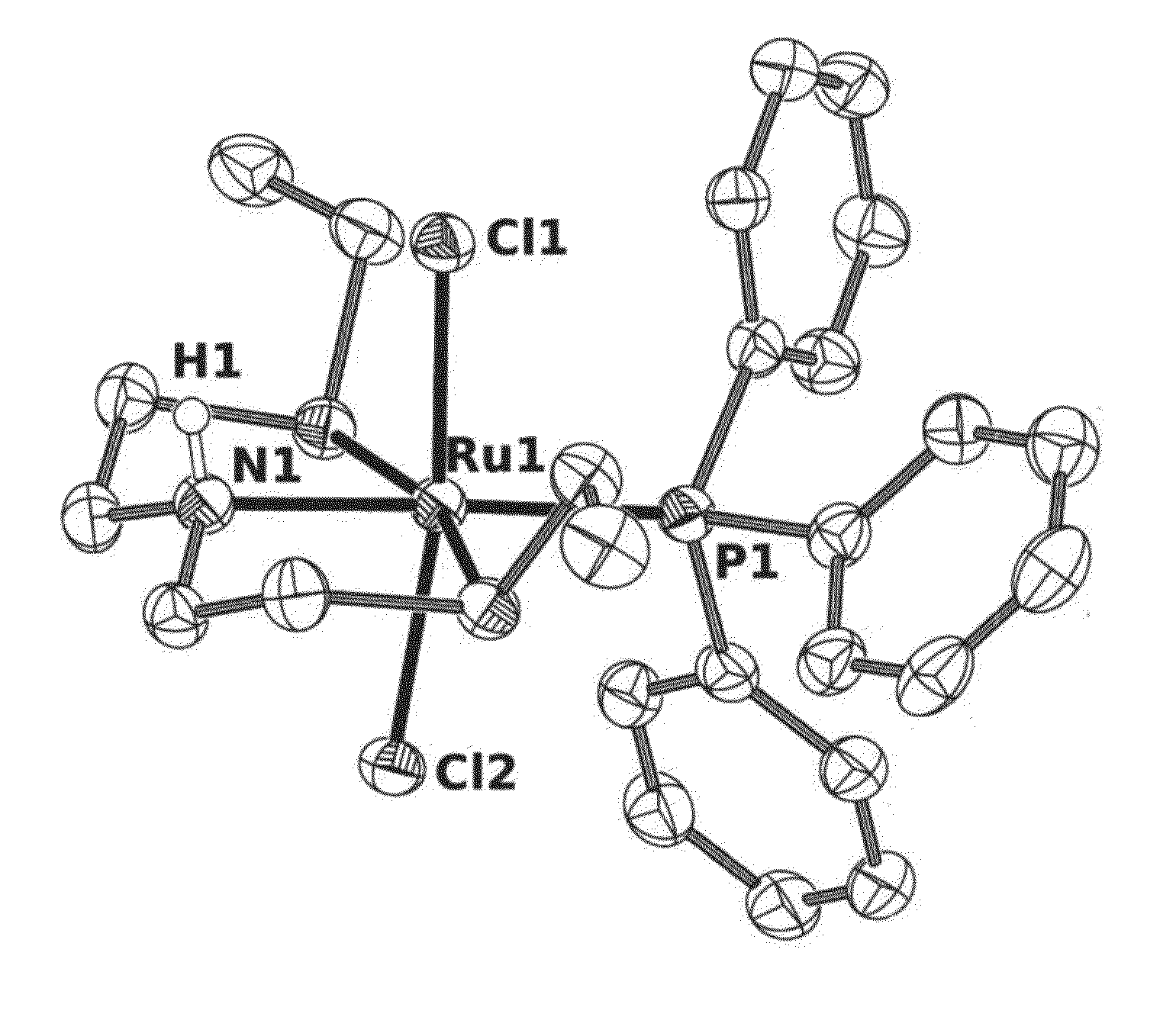
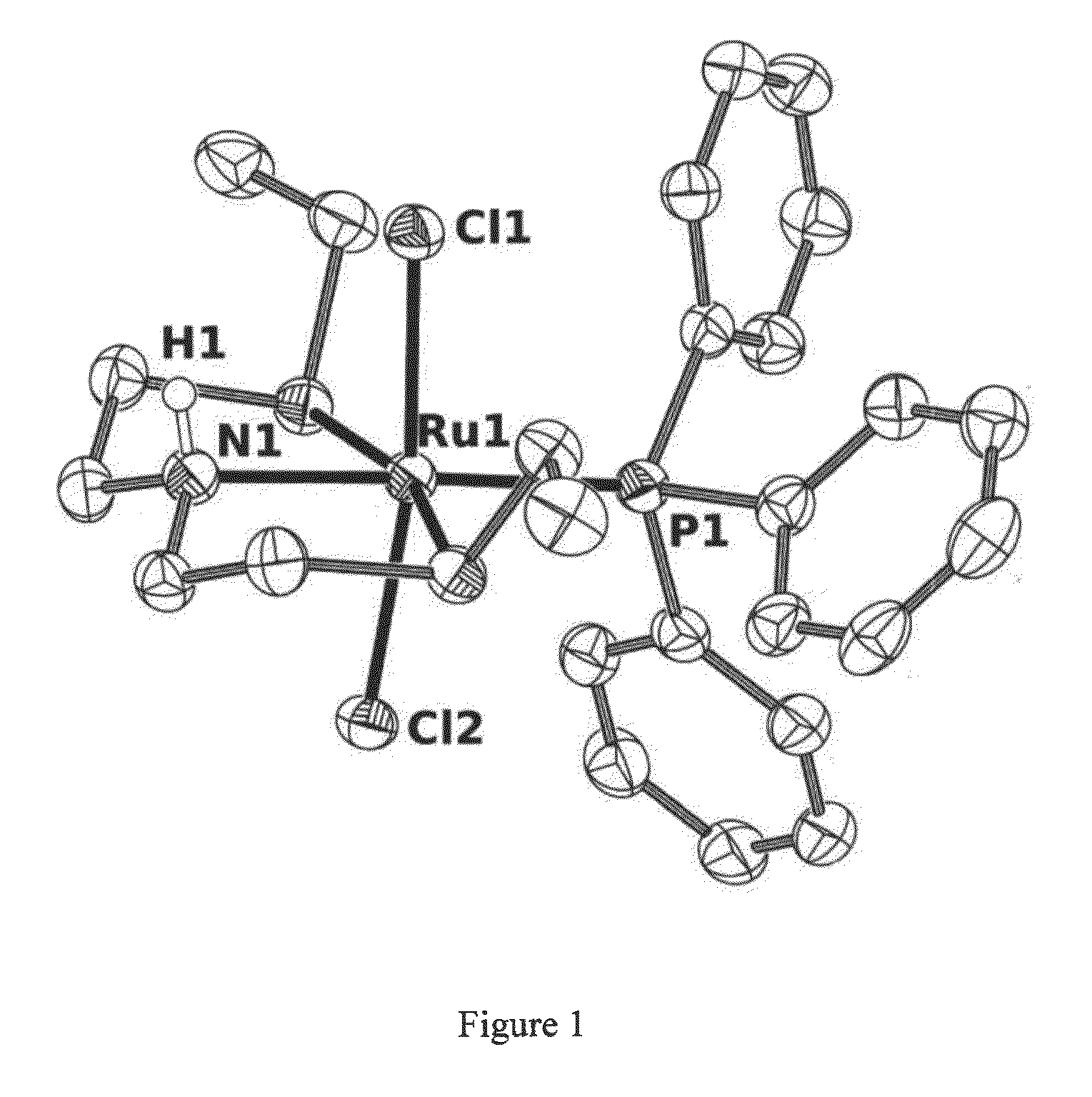
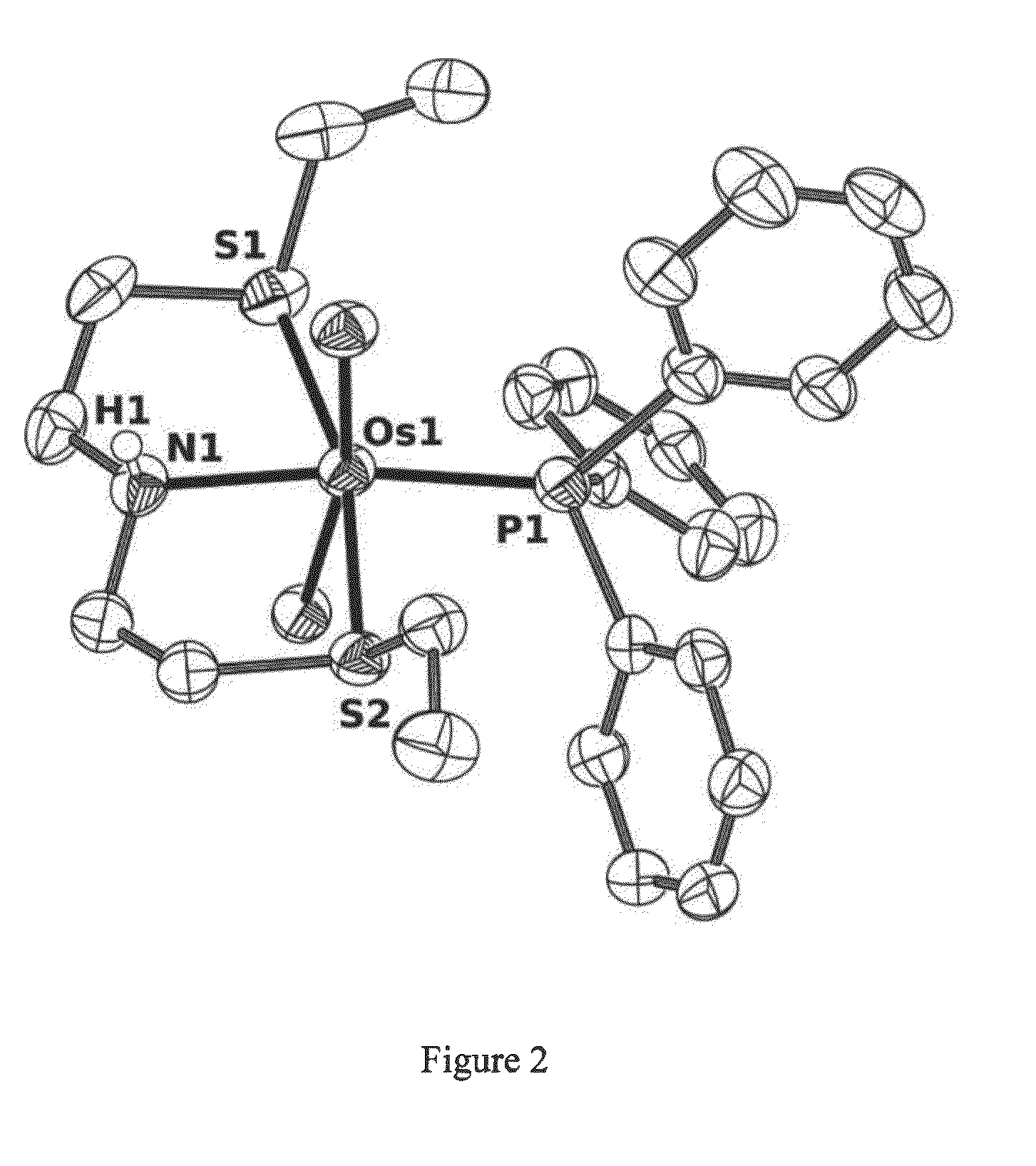
Catalysts based on Amino-Sulfide Ligands for Hydrogenation and Dehydrogenation Processes
a technology of aminosulfide ligands and hydrogenation and dehydrogenation, applied in the field of catalysts, can solve the problems of dangerous exothermic hydride reduction methods, large amount of chemical waste, inconvenient and expensive use of hydride reducing reagents,
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Synthesis of EtS(CH2)2NH2
[0073]All manipulations were carried in air. In a 250 mL flask, 24 g (0.191 mol) of NaOH was dissolved in 100 mL of CH3OH and EtSH (15 g, 0.242 mol) was added. After 10 minutes of reaction, 28.07 g (0.242 mol) of 2-chloroethylamine hydrochloride was slowly added to the mixture and the reaction was left to stir overnight. In the following morning, methanol was removed in vacuo and the remaining semisolid was extracted with 3×30 mL of Et2O. The obtained extract was filtered through a short plug (2 cm×1 cm) of Al2O3 and ether was removed under reduced pressure (15 mm Hg) to yield a pale yellow oil (23.4 g, 92%)
[0074]1H NMR ([D6]Benzene) δ=2.54 (t, J=6.2 Hz, 2H, CH2), 2.25 (t, J=6.2 Hz, 2H, CH2), 2.18 (q, J=7.4 Hz, 2H, SCH2), 1.02 (t, J=7.4 Hz, 3H, CH3), 0.76 (br, 211, NH2). 13C{1H} NMR ([D6]Benzene) δ=41.72 (s, 1C, NCH2), 36.22 (s, 1C, CH2S), 25.72 (s, 1C, SCH2), 15.11 (s, 1C, CH3).
example 2
Synthesis of (EtS(CH2)2)2NH
[0075]All manipulations were carried in air. To a solution of bis(2-chloroethyl)amine hydrochloride (23.9 g, 0.134 mol) in 80 mL of methanol were added NaOH (6.27 g, 0.157 mol) and sodium ethanethiolate (25 g, 0.298 mol). The resulting mixture was stirred overnight at room temperature. After that time, methanol was removed in vacuo. The remaining yellow slurry was extracted with 3×15 mL of Et2O and the obtained extract was filtered through a short plug (2 cm×1 cm) of Al2O3. The solvent was removed in vacuo to yield a pale yellow oil (23.0 g, 89%).
[0076]Alternatively, (EtS(CH2)2)2NH was prepared by the following, slightly different method. Again all manipulations were performed in air. To a solution of NaOH (6.27 g, 0.157 mol) and sodium ethanethiolate (25 g, 0.298 mol) in 80 mL of methanol was slowly added bis(2-chloroethyl)amine hydrochloride (23.9 g, 0.134 mol) in 65 ml of methanol. The mixture was stirred overnight at room temperature, and then the solv...
example 3
Synthesis of(EtSC2H4)2O
[0078]All manipulations were performed in air. To a solution of NaSEt (5.41 g, 64.4 mmol) in 40 mL of methanol was added a solution of bis(2-chloroethyl) ether (4.6 g, 32.2 mmol) in 20 mL of methanol. The mixture was stirred overnight at 40° C., and then the solvent was removed under reduced pressure. The resulting slurry was extracted with 2×20 mL of hexanes and the obtained extract was filtered through a short plug (2×1 cm) of basic alumina. The solvent was evaporated to give a transparent oil (4.07 g, 64%). 1H NMR ([D6]Benzene) 8 3.39 (t, J=6.9 Hz, 4H, OCH2), 2.52 (t, J=6.9 Hz, 4H, CH2S), 2.31 (q, J=7.4 Hz, 4H, SCH2), 1.05 (t, J=7.4 Hz, 6H, CH3). 13C{1H} NMR ([D6]Benzene) S 71.38 (s, 2C, CH2O), 31.50 (s, 2C, CH2S), 26.66 (s, 2C, SCH2), 15.13 (s, 2C, CH3).
PUM
| Property | Measurement | Unit |
|---|---|---|
| Pressure | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More