Modulators of the beta-3 adrenergic receptor useful for the treatment or prevention of disorders related thereto
a beta-3 adrenergic receptor and receptor technology, applied in the direction of cardiovascular disorders, organic chemistry, drug compositions, etc., can solve the problems of increased tachycardia, accelerated mortality, and initial improvement in contractility of these agents, so as to improve contractile function and hemodynamic status, inhibit the effect of contractility
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
of Compounds of the Present Invention
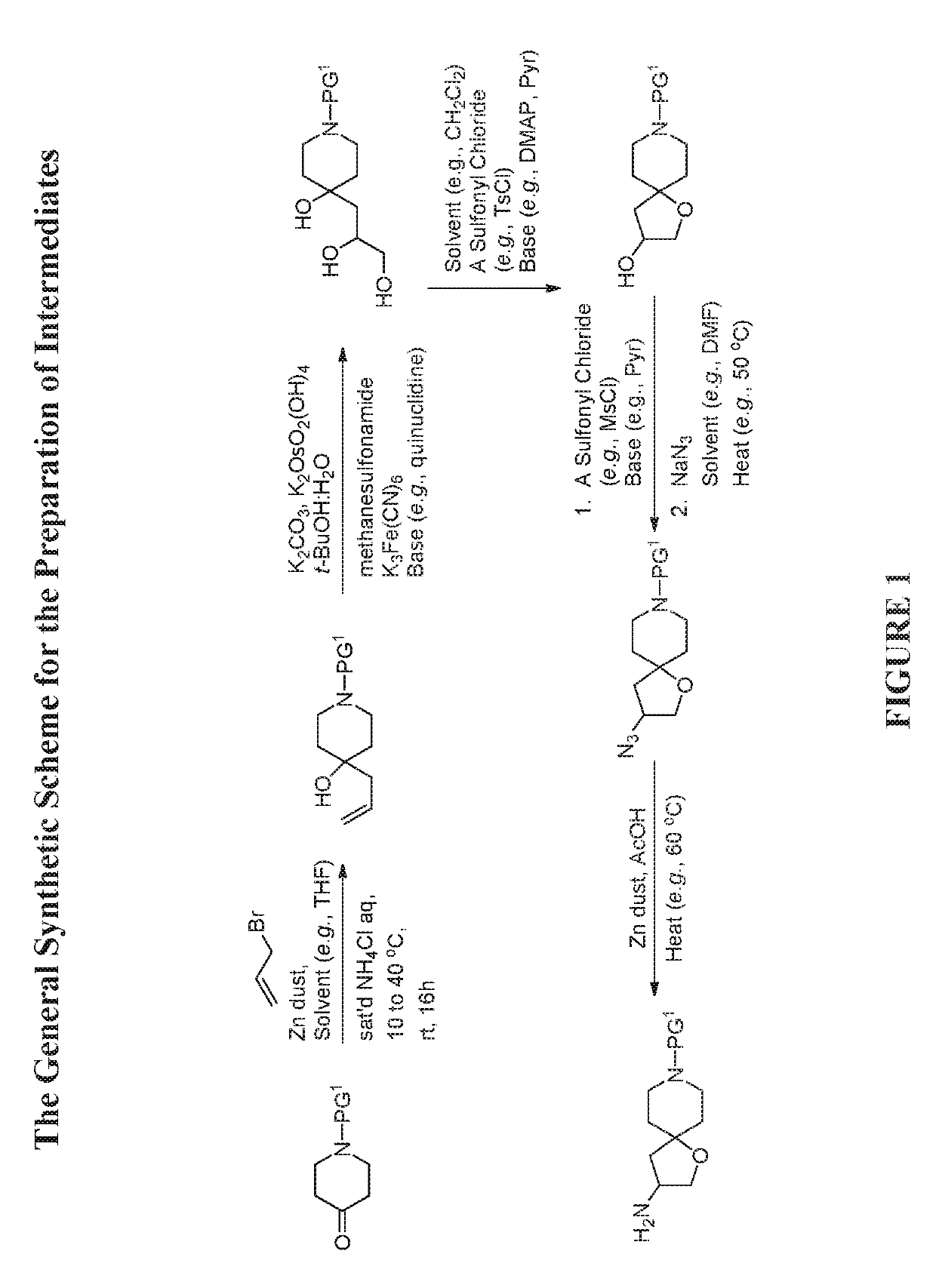
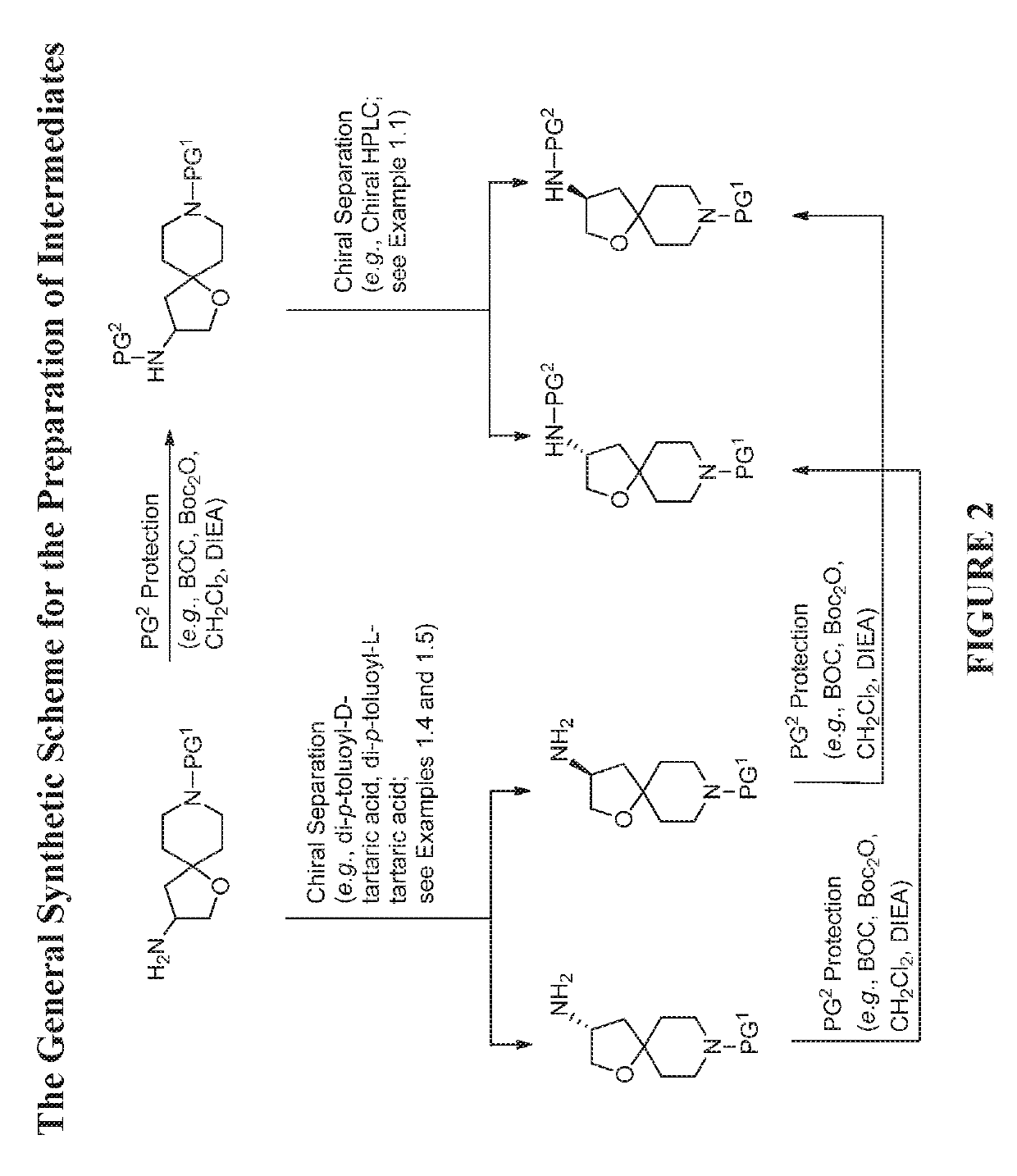
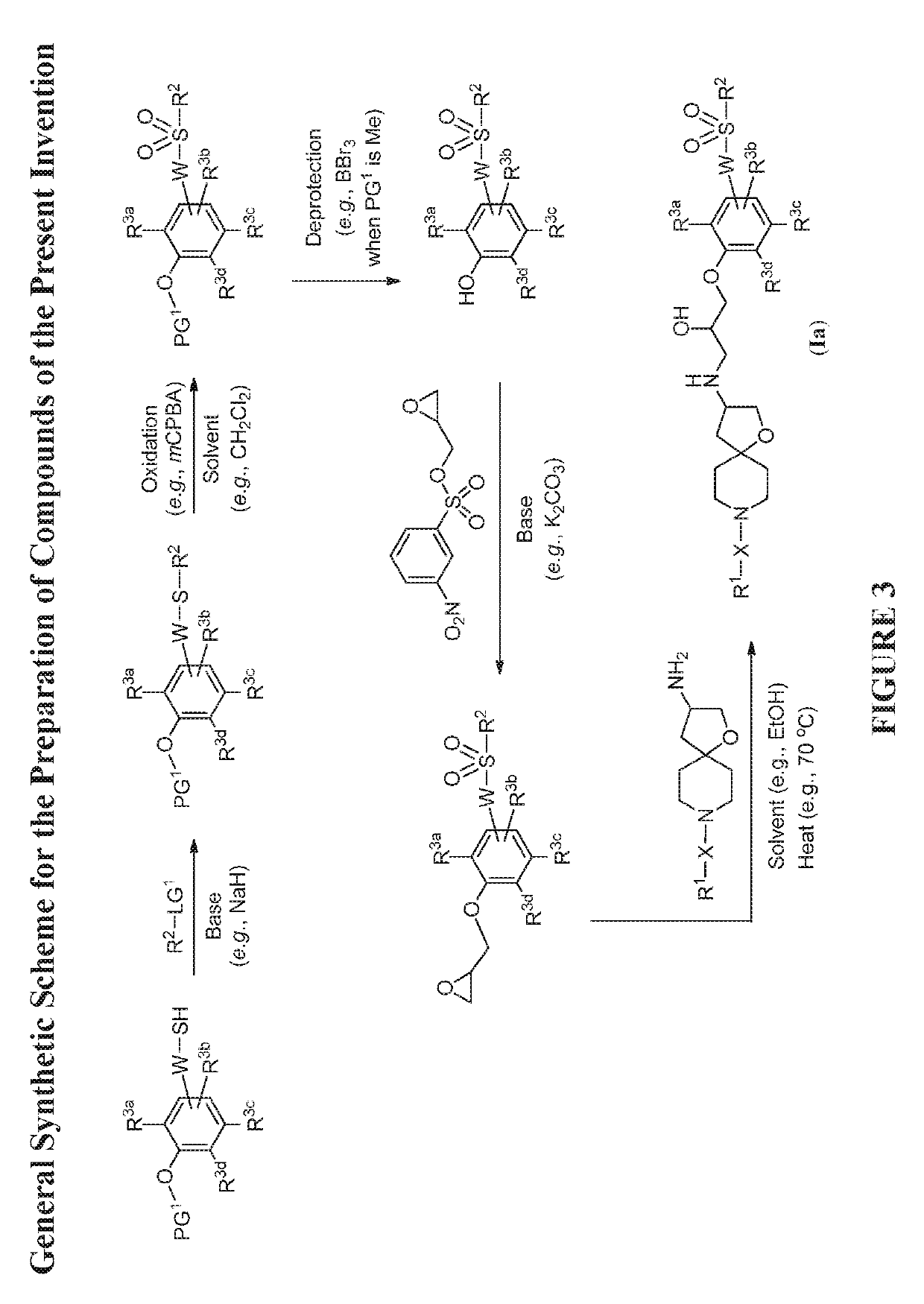
[0584]The compounds disclosed herein and their syntheses are further illustrated by the following examples. Additional illustrated syntheses for compounds of the present invention are shown in FIGS. 1 to 24 where the symbols have the same definitions as used throughout this disclosure. The following examples are provided to further define the invention without, however, limiting the invention to the particulars of these examples. The compounds described herein, supra and infra, are named according to the AutoNom version 2.2, CS ChemDraw Ultra Version 9.0.7, or ChemBioDraw Ultra 12.0.2.1076. In certain instances common names are used and it is understood that these common names would be recognized by those skilled in the art.
[0585]Chemistry: Proton nuclear magnetic resonance (1H NMR) spectra were recorded on a Bruker Avance III-400 equipped with a 5 mm BBFO probe. Chemical shifts are given in parts per million (ppm) with the residual solvent signa...
example 1.1
of Benzyl 3-((tert-Butoxycarbonyl)amino)-1-oxa-8-azaspiro[4.5]decane-8-carboxylate
Step A: Preparation of Benzyl 4-Allyl-4-hydroxypiperidine-1-carboxylate
[0587]To a mixture of benzyl 4-oxopiperidine-1-carboxylate (51 g, 218.6 mmol) in THF (36.44 mL) were added 3-bromoprop-1-ene (54.72 mL, 655.92 mmol) and saturated NH4Cl (114 mL, 218.6 mmol) aqueous solution. Then Zinc dust (31.59 g, 483.1 mmol) was added portion wise while the internal reaction temperature was kept below 40° C. The reaction was stirred at room temperature overnight. After the reaction was completed, it was quenched with H2SO4 (10%, 225 mL). The reaction mixture was filtered through a pad of Celite® and washed with MTBE (1 L). The aqueous layer was extracted with MTBE (2×) and EtOAc (lx). The combined organic layers were washed with water and brine, and then dried over MgSO4, filtered and concentrated to give the title compound (62.39 g, 104% yield). This material was used in the next step without further purificatio...
example 1.2
of (S)-3,3,3-Trifluoro-2-methoxy-N—((S)-8-(naphthalen-2-ylsulfonyl)-1-oxa-8-azaspiro[4.5]decan-3-yl)-2-phenylpropanamide and (R)-3,3,3-Trifluoro-2-methoxy-N—((S)-8-(naphthalen-2-ylsulfonyl)-1-oxa-8-azaspiro[4.5]decan-3-yl)-2-phenylpropanamide
Step A: Preparation of one Enantiomer of tert-Butyl (8-(Naphthalen-2-ylsulfonyl)-1-oxa-8-azaspiro[4.5]decan-3-yl)carbamate (from 1st Enantiomer)
[0614]The 1st enantiomer of benzyl 3-((tert-butoxycarbonyl)amino)-1-oxa-8-azaspiro[4.5]decane-8-carboxylate (1.13 g, 2.89 mmol) from chiral HPLC in Example 1.1 was dissolved in MeOH (10 mL).
[0615]Palladium / C (30.72 mg, 0.289 mmol) and a H2 balloon were applied. The reaction was stirred at room temperature overnight at room temperature. The next day, the H2 balloon was removed. The reaction mixture was filtered through a pad of Celite®, washed with EtOAc and MeOH, and concentrated to give an enantiomer of tert-butyl 1-oxa-8-azaspiro[4.5]decan-3-ylcarbamate (0.64 g, 86% yield) as a colorless gum which was ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Molar density | aaaaa | aaaaa |
| Molar density | aaaaa | aaaaa |
| Molar density | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More