Method for preparing 11-methoxyl-3,7,11-trimethyl-2,4-dodecadienoate
A technology of isopropyl carbadienoate and ammonium carbadienoate, which is applied in the field of preparation of insect growth regulators, can solve the problems of high cis-ester content, affecting product performance, unqualified and the like, avoiding hydrolysis, The effect of high pass rate
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0008] The preparation method steps of 11-methoxy-3,7,11-trimethyl-2,4-dodecadienoic acid isopropyl ester of the present embodiment are as follows:
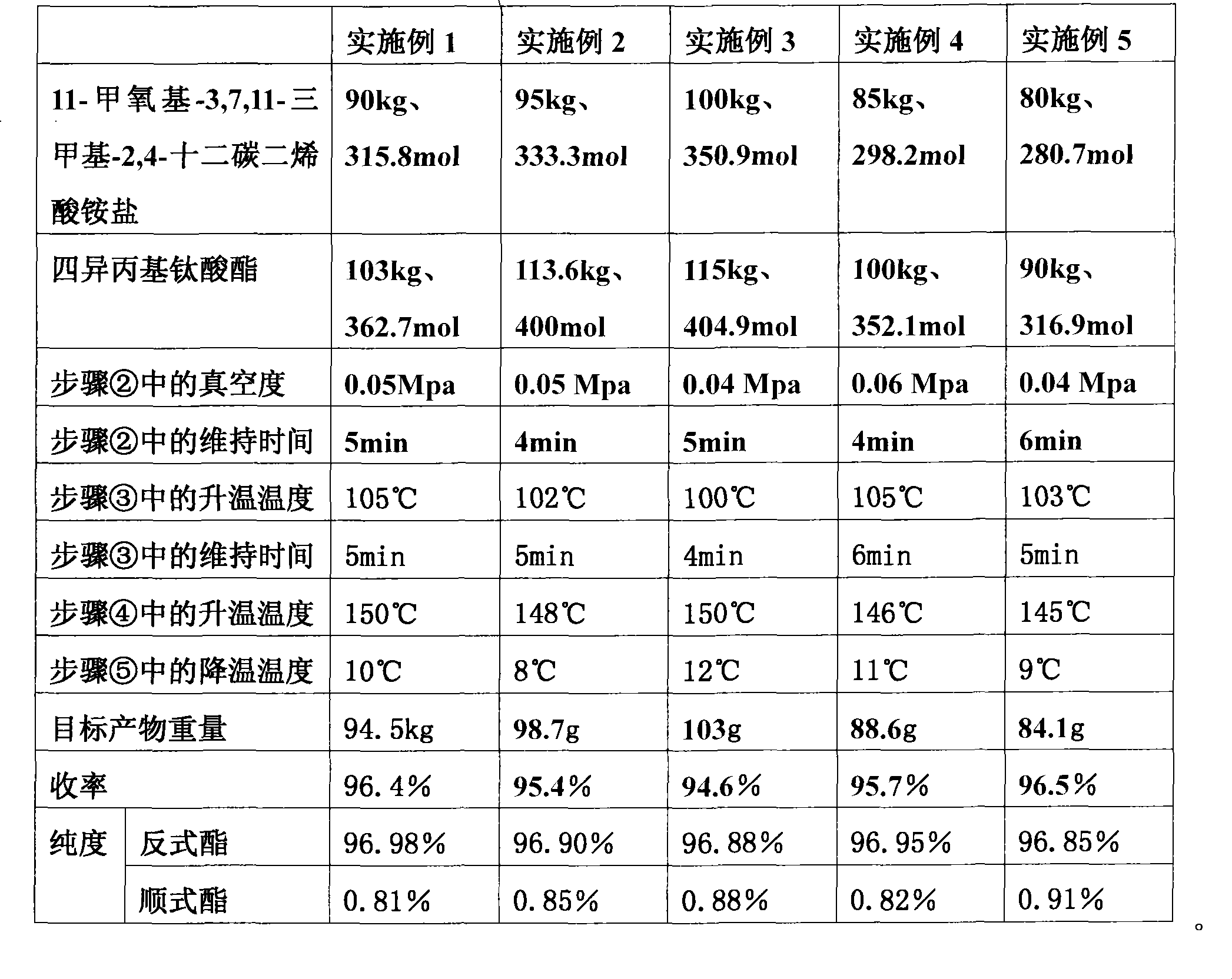
[0009] ① Under a nitrogen atmosphere and stirring, put 54kg of n-hexane and 90kg of 11-methoxyl-3,7,11-trimethyl-2,4-dodecane into a reactor with a reflux condenser successively Dienoic acid ammonium salt (315.8 mol) and 103 kg of tetraisopropyl titanate (362.7 mol). Among them, n-hexane and tetraisopropyl titanate are industrial-grade raw materials.
[0010] ② Turn on the vacuum and keep the vacuum for 5 minutes after reaching 0.05Mpa.
[0011] ③Raise the temperature slowly to 105°C within 25-30 minutes, and maintain at this temperature for 5 minutes to remove ammonia gas.
[0012] ④Raise the temperature to 150°C within 8 to 12 minutes, turn off the vacuum, and keep the temperature at this temperature for 1 hour to complete the reaction.
[0013] 5. the reaction product is rapidly reduced to 10 DEG C, and 53kg of normal hexan...
Embodiment 2~ Embodiment 5
[0015] The preparation method of each embodiment is basically the same as that of Example 1, and the differences are shown in Table 1.
[0016]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More