Human interferon alpha derivative and polyethylene glycol modified substance thereof
A technology of PEGylation and interferon α, which is applied in the field of biomedicine, can solve the problems affecting the purity of PEGylation modifications and the low rate of PEGylation modification, and achieve good clinical application prospects, good water solubility, The effect of low antigenicity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0034] Example 1: gly-gly-gly-gly-gly-IFNα-2a (hereinafter referred to as Gly(5)-IFN
[0035] α) Secreted expression in Pichia pastoris
[0036] 1. Design and acquisition of the target gene:
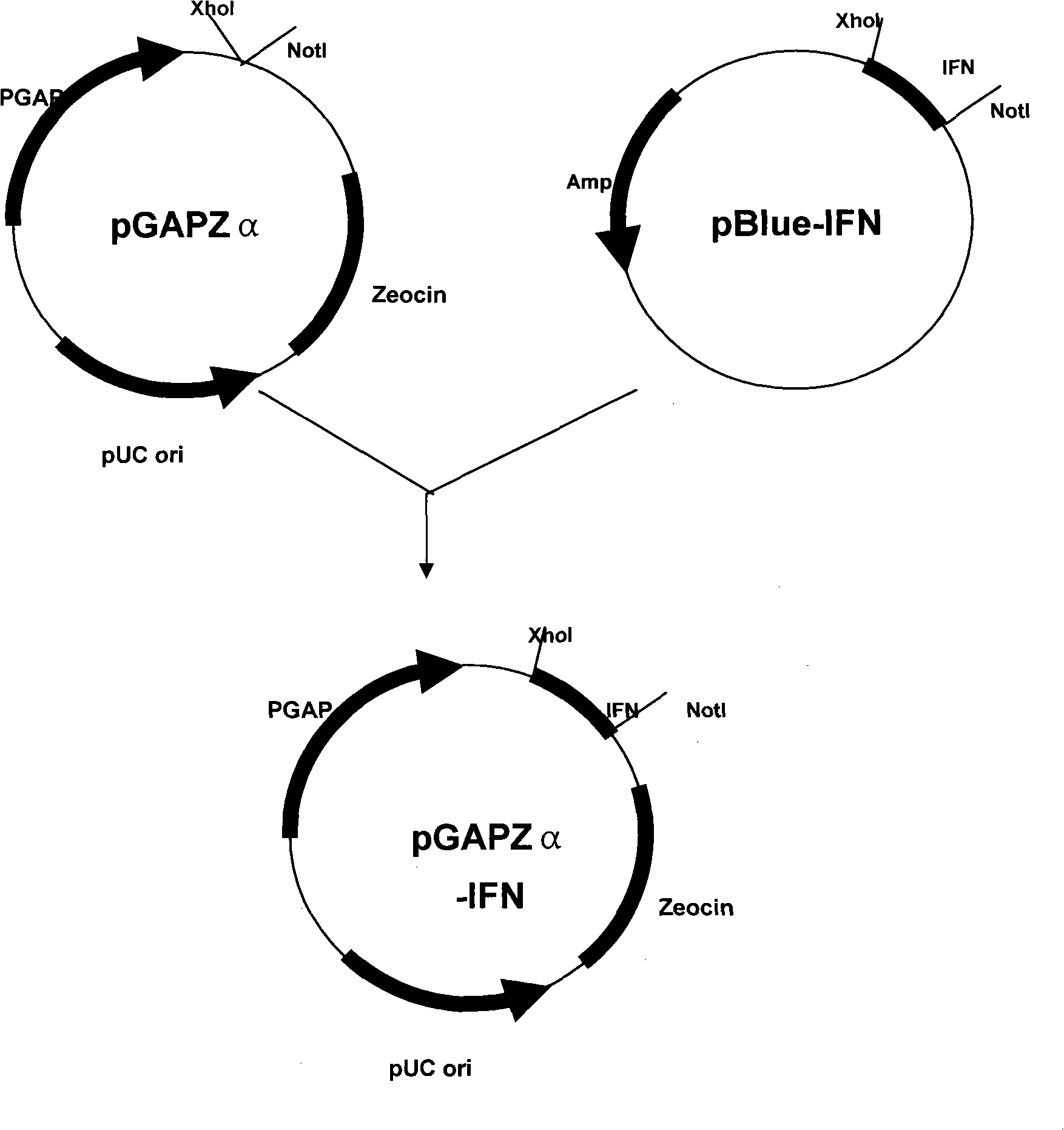
[0037] The amino acid sequence of IFNα-2a is shown in SEQ ID No.1. After obtaining the cDNA of IFNα-2a through Genebank, the corresponding codons were changed to yeast preference, and the corresponding nucleotide sequence of gly-gly-gly-gly-gly was added to the N-terminus. This CDNA sequence was used in the construction of the pGAPZα expression plasmid (purchased from invitrogen) of Pichia pastoris GS115 (purchased from invitrogen). Secreted expression was achieved after transformation of GS115 host bacteria. Therefore, the enzyme recognition site CTC GAG AAA AGA of KEX2 was added in the design, wherein CTC GAG is the XhoI restriction site. At the same time, a double stop codon TGA TAA and a NotI restriction sequence GCG GCCGC were introduced into the 3' end. The cDNA sequence of IF...
Embodiment 2
[0054] Example 2: Purification of Gly(5)-IFNα
[0055] The first step: cationic gel column (CM Sepharose F.F. gel purchased from Amersham Biosciences) chromatography:
[0056] Acetate buffer with pH 3.8-4.6 was used for column loading and elution, and electrophoresis monitoring was used to collect target objects. Then, the target substance was dialyzed with a pH 7.5-8.5 Tris-HCl buffer solution.
[0057] The second step: anion gel column (DEAE Sepharose F.F. gel purchased from Amersham Biosciences) chromatography:
[0058] The pH7.5-8.5 Tris-HCl buffer solution is used for column loading and elution, and the target substance is collected. Then dialyze the target substance with pH 7.5-8.5 phosphate buffer solution.
Embodiment 3
[0059] Example 3: Preparation and purification of PEG-coupling modified samples
[0060] 1. Dialyze the Gly(5)-IFNα sample obtained in Example 1 with phosphate buffer (pH 6.0), and then add ALD-PEG 40KD (purchased from Beijing Jiankai Technology Co., Ltd.) with a molar ratio of 1:3 The modification is carried out at 2-15°C, and the reaction time is 40 hours. The obtained modified sample was tested by SDS-PAGE. The test results showed that after PEG coupling modification, the molecular weight increased from the original 19,000 Daltons to nearly 90,000 Daltons, and the modification rate reached about 60%, and the target compound was obtained mPEG-Gly(5)-IFNα.
[0061] 2. Purification of mPEG-Gly(5)-IFNα:
[0062] The mPEG-Gly(5)-IFNα was adjusted with acetate buffer (pH 4.0-5.0) to terminate the reaction, and purified on a cationic gel column SP Sepharose F.F. gel column. Elute with NaCl (0.15M) solution to collect the target substance with a purity of over 95%. The result i...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com