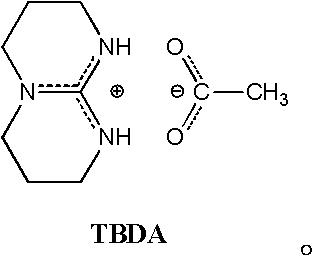
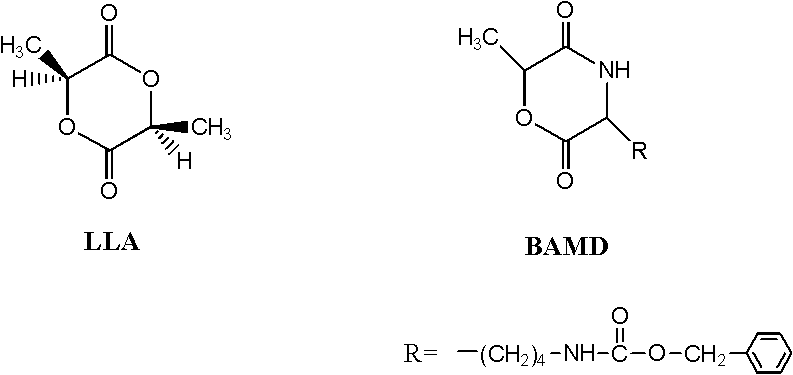
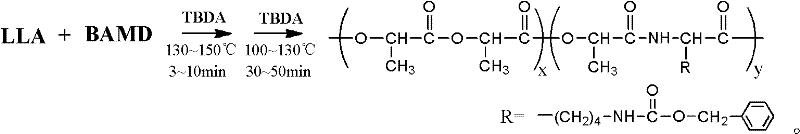
Process method for synthesizing lactic acid-lysine copolymer by catalytically opening loop and copolymerizing with acetic bicyclo-guanidine
A lysine copolymer and bicyclic guanidine catalyzed lactide technology, which is applied in the direction of medical preparations, drug combinations, and pharmaceutical formulations of non-active ingredients, can solve problems such as potential safety hazards and poor catalytic effects, and achieve rapid response , high biological safety, and high conversion rate of monomer
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0031] Add 1.000g (6.944mmol) of monomer L-lactide and 0.0473g (0.142mmol) of L-lactide lysine to the reaction kettle to control the molar ratio of L-lactide and morpholine lysine Be: 98 / 2; Add catalyst bicyclic guanidine acetate 0.0141g (0.071mmol), control the molar weight sum of comonomer lactide and lysine morpholine diketone and the ratio of the molar weight of catalyst bicyclic guanidine acetate is: 100 / 1. After three times of evacuation-argon operation, the reactor was heated to 130° C. for 5 minutes under vacuum, and then continued to react at 100° C. for 40 minutes.
[0032] 20ml of dichloromethane was used to dissolve the polymer in a pressure test tube. Then 5 ml of triethylsilane, 0.1 ml of triethylamine and 0.15 g of palladium dichloride were added. Reaction at room temperature for 72h to remove the protecting group O-benzyl. After the reaction, palladium dichloride was filtered off, and 5 ml of methanol was added to the filtrate to completely react with unreac...
Embodiment 2
[0035]Add 1.000g (6.944mmol) of monomer L-lactide and 0.1480g (0.443mmol) of L-lactide lysine to the reaction kettle to control the molar ratio of L-lactide and morpholine lysine Be: 94 / 6; Add catalyst bicyclic guanidine acetate 0.0147g (0.074mmol), control the molar weight sum of comonomer lactide and lysine morpholine diketone and the ratio of the molar weight of catalyst bicyclic guanidine acetate is: 100 / 1. After the reaction kettle was subjected to vacuum pumping and argon filling operations for three times, it was heated to 140° C. for 4 minutes under vacuum, and then continued to react at 120° C. for 30 minutes.
[0036] 20ml of dichloromethane was used to dissolve the polymer in a pressure test tube. Then 5 ml of triethylsilane, 0.1 ml of triethylamine and 0.15 g of palladium dichloride were added. Reaction at room temperature for 72h to remove the protecting group O-benzyl. After the reaction, palladium dichloride was filtered off, and 5 ml of methanol was added to...
Embodiment 3
[0039] Add 1.000g (6.944mmol) of monomer L-lactide and 0.2577g (0.772mmol) of L-lactide and 0.2577g (0.772mmol) of L-lactide to the reaction kettle to control the molar ratio of L-lactide and morpholine lysine Be: 90 / 10; Add catalyst bicyclic guanidine acetate 0.0154g (0.077mmol), control the molar weight sum of comonomer lactide and lysine morpholine diketone and the ratio of the molar weight of catalyst bicyclic guanidine acetate is: 100 / 1. After three times of evacuation-argon operation, the reactor was heated to 150° C. for 3 minutes under vacuum, and then continued to react at 130° C. for 20 minutes.
[0040] 20ml of dichloromethane was used to dissolve the polymer in a pressure test tube. Then 5 ml of triethylsilane, 0.1 ml of triethylamine and 0.15 g of palladium dichloride were added. Reaction at room temperature for 72h to remove the protecting group O-benzyl. After the reaction, palladium dichloride was filtered off, and 5 ml of methanol was added to the filtrate ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight distribution | aaaaa | aaaaa |
| molecular weight distribution | aaaaa | aaaaa |
| molecular weight distribution | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com