Medicine for controlling swine erysipelas
A porcine erysipelas and drug technology, applied in the field of medicine and medical engineering, can solve problems such as immune failure, imprecise formula, and unsatisfactory control effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0014] Test time: August 2010.
[0015] Test site: Shensancheng Pig Farm, Cheng'an County, Hebei Province.
[0016] Pigs to be tested: 80-105 days old, weighing 25-50kg, 116 Yorkshire commercial generation pigs, due to reasons such as lack of vaccination, hot weather, and humid pig houses, outbreaks of porcine erysipelas, and the same house and the same group.
[0017] Test drug: pure Chinese herbal medicine preparation of the present invention, each raw material drug is purchased from Dongfang Medicine City, Anguo City, Hebei Province, and its composition is expressed in parts by mass: 5 parts of honeysuckle, 5 parts of Daqingye, and 5 parts of Zihua Diding , 5 parts of comfrey, 4 parts of Guanzhong, 4 parts of Hanshuishi, 4 parts of paeonol, 4 parts of Anemarrhena, 3 parts of Scrophulariaceae, 3 parts of Selaginella, 3 parts of red peony, 3 parts of bellflower, 2 parts of rhubarb, gardenia 2 parts of licorice and 2 parts of licorice. The above-mentioned Chinese herbal medi...
Embodiment 2
[0031] Test time: July 2011.
[0032] Place for the test: Li Zhiguo pig farm in Wei County, Hebei Province.
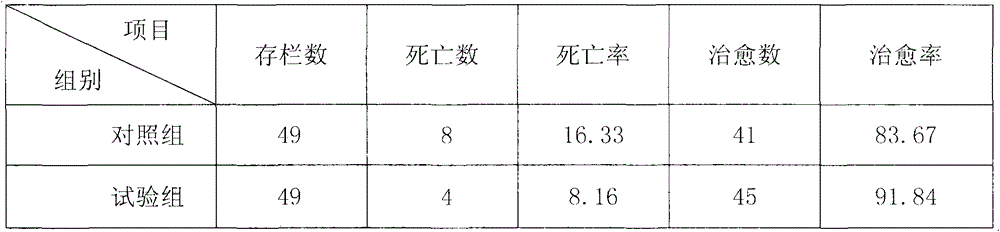
[0033] Pigs to be tested: 73-112 days old, weight 23-52kg, 98 Changbai commercial generation pigs, outbreaks of erysipelas disease due to reasons such as lack of immunization, sweltering weather, and unclean pig houses, all in the same group.
[0034] Drug for testing: namely the present invention, the procurement of raw materials is the same as in Example 1, and its composition is expressed in parts by mass: 15 parts of Flos Lonicerae Flos, 15 parts of Folium Folium, 15 parts of Zihua Diding, 15 parts of Comfrey, 12 parts of Rhizome, 12 parts of Hanshuishi, 12 parts of paeonol, 12 parts of Anemarrhena, 10 parts of Scrophulariae, 10 parts of Selaginella, 10 parts of red peony, 10 parts of bellflower, 6 parts of rhubarb, 6 parts of gardenia, and 6 parts of licorice. Processing method is with embodiment 1.
[0035] Test group: 49 heads, treated with the medicine of the...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More