mpeg-mal-aGLP-1 composite
A technology of mpeg-mal-aglp-1 and complexes, applied in the field of monomethoxypolyethylene glycol-human glucagon-like peptide-1 derivative complexes, can solve the problem of polyethylene glycol-modified protein/ Difficult to control the site and degree of modification of the polypeptide, difficult to purify and quality control, cumbersome preparation steps, etc., to achieve the effect of low cost, simple purification and high yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0028] Preparation of embodiment 1mPEG-MAL-aGLP-1 complex
[0029] In this embodiment, the structural formula of the aGLP-1 derivative is: aGLP-1(7-36), namely Seq ID No.2, and the sequence of Seq ID No.2 is as follows:
[0030] His-Xaa2-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Val-Ser-Ser-Tyr-Leu-Glu-Gly-Gln-Ala-Ala-Xaa20-Glu-Phe-Ile-Ala-Trp- Leu-Val-Xaa28-Gly-Xaa30.
[0031] Where Xaa2=D-Ala, Xaa20=Ser, Xaa28=Ala, Xaa30=Cys, and the molecular weight of mPEG-MAL is 2KD.
[0032] In the first step, 1.0 mg of aGLP-1 was dissolved in 0.5 ml of 100 mM, pH 7.4 phosphate buffer, and then mixed with 0.5 ml of 200 mM phosphate buffer of pH 6.0 to 9.0;
[0033] In the second step, add 0.6 mg of mPEG-MAL with a molecular weight of 2KD to the solution in the first step, and mix evenly;
[0034] In the third step, the solution obtained in the second step is placed at 4°C to 30°C for mPEG modification reaction for 0.5 to 24 hours;
[0035] In the fourth step, the solution treated in the third step...
Embodiment 2
[0041] Preparation of embodiment 2mPEG-MAL-aGLP-1 complex
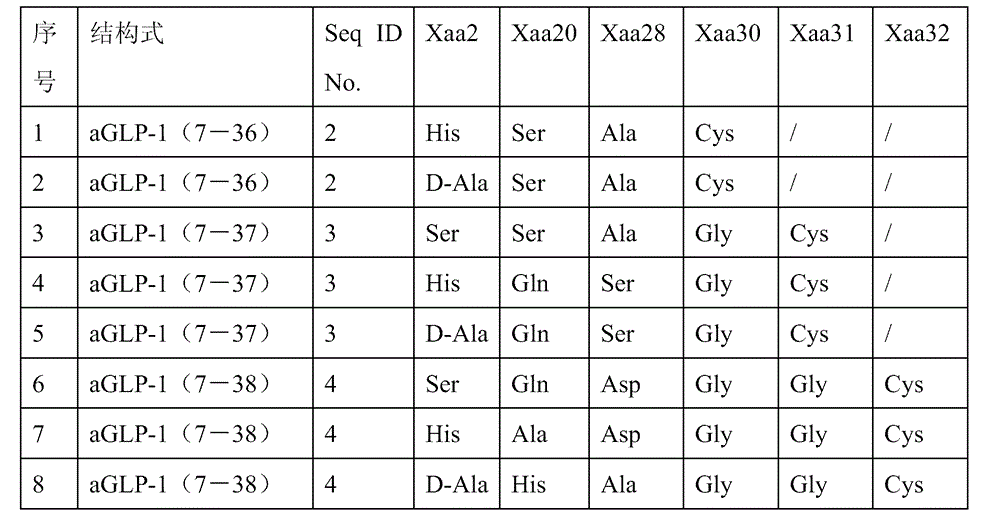
[0042] In this example, the structural formula of the aGLP-1 derivative used is shown in Table 1, and the molecular weight of mPEG-MAL is 2KD.
[0043] In this example, the mPEG-MAL-aGLP-1 complex was prepared according to the experimental conditions and procedures in Example 1.
[0044] Table 1aGLP-1 derivative structure
[0045]
Embodiment 3
[0046] Embodiment 3 Preparation of mPEG-MAL-aGLP-1 complex
[0047] In this example, the structural formula of the aGLP-1 derivative is: aGLP-1(7-36), namely Seq ID No.2, wherein Xaa2=Ser, Xaa20=Ser, Xaa28=Ala, Xaa30=Cys, mPEG- The molecular weight of MAL is 20KD.
[0048] In the first step, 1.0 mg of aGLP-1 was dissolved in 0.5 ml of 100 mM, pH 7.4 phosphate buffer, and then mixed with 0.5 ml of 200 mM phosphate buffer of pH 6.0 to 9.0;
[0049] In the second step, add 6.0 mg of mPEG-MAL with a molecular weight of 20KD to the solution in the first step, and mix evenly;
[0050] The third step to the fifth step are carried out according to the corresponding operation steps in the first embodiment.
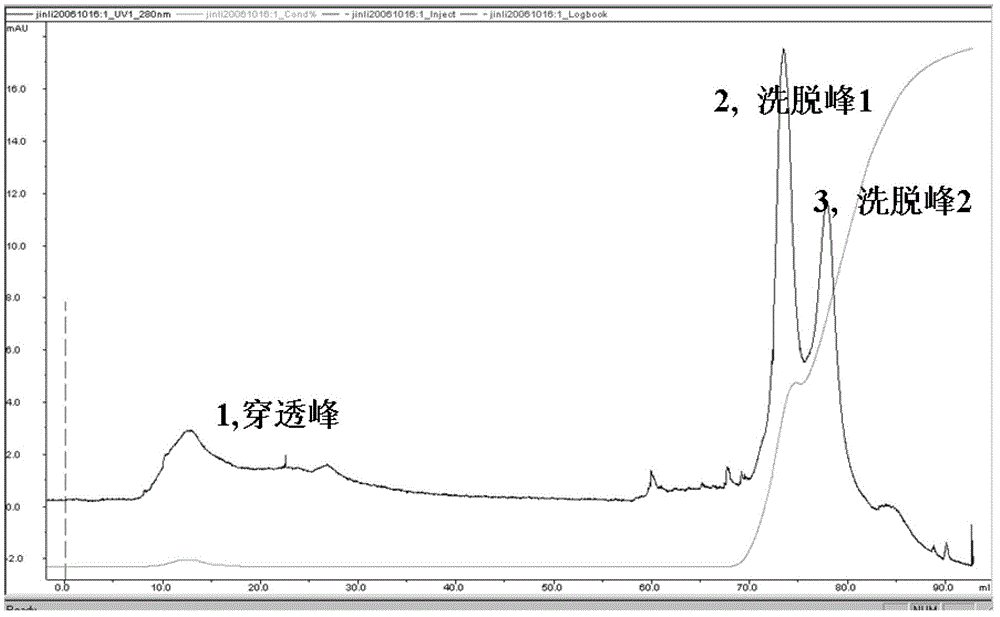
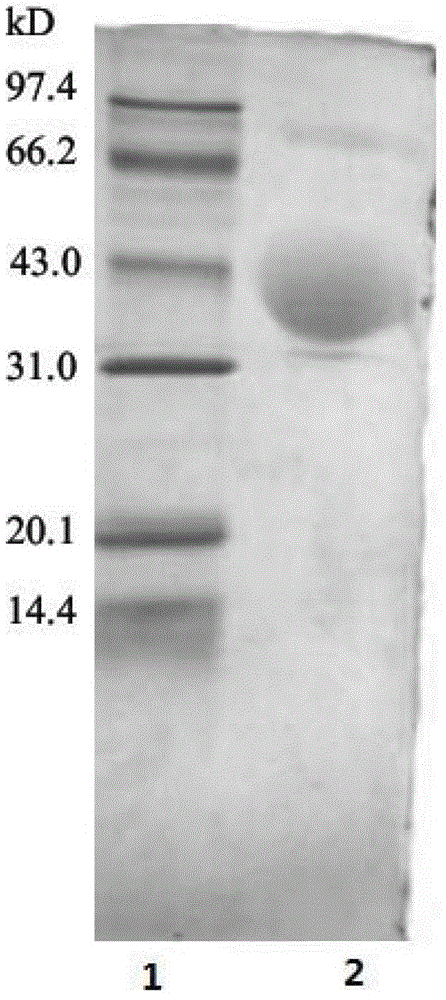
[0051] In the sixth step, the eluate collected in the fifth step was concentrated with a Millipore Amicon Ultra-15 ultrafiltration tube with a molecular weight cut-off of 1 to 5 KD, and freeze-dried to obtain 0.7 mg of aGLP-1 derivative with a molecular weight of 23.5 KD pure co...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Molecular weight | aaaaa | aaaaa |
| Molecular weight | aaaaa | aaaaa |
| Molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More