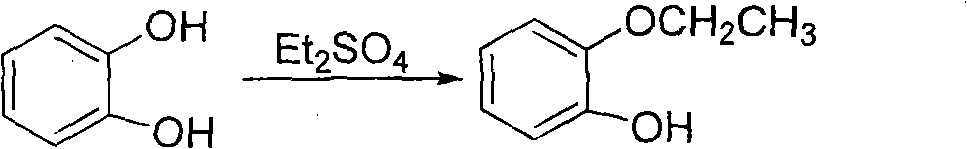
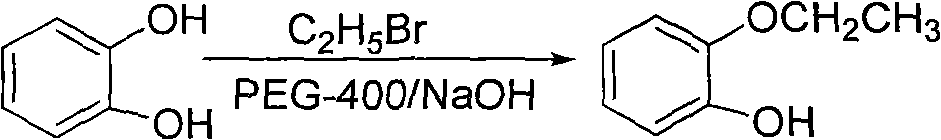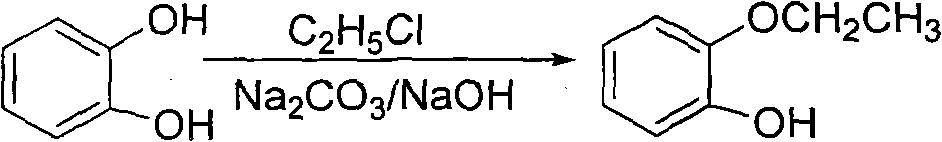Method for preparing 2-(2-ethoxyphenoxy) ethyl bromide
A technology of ethoxyphenoxy and ethoxyphenol, which is applied in the field of purification of the intermediate by acid-base back extraction and recrystallization, the preparation and purification of the intermediate 2-ethyl bromide, to improve the purity , avoid the vacuum distillation process, and optimize the preparation process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0045] Under nitrogen protection, dissolve 100g of catechol in 230ml of water. After dissolving at room temperature, slowly add 20% aqueous sodium hydroxide solution dropwise to adjust the pH value to 8-9, then add 14.5g of tetrabutylammonium bromide, and heat up To 70 DEG C, begin to drop slowly 145ml diethyl sulfate (completely dropwise in about 4 hours), add 20% sodium hydroxide solution at any time during the dropping process, keep the pH of the reaction solution at 9-10, and keep The temperature of the reaction solution was 75-80° C., and the dropwise addition was completed (thin-layer chromatography showed that the raw material disappeared).
[0046] The reaction solution was lowered to room temperature, and concentrated hydrochloric acid was added dropwise to adjust the pH value of the reaction solution to 2-3, extracted twice with toluene (400ml×2), the organic phases were combined, and washed with 400ml saturated sodium chloride and 400ml distilled water respectively. ...
Embodiment 2
[0048]Under nitrogen protection, dissolve 100g of catechol in 230ml of water. After dissolving at room temperature, slowly add 20% aqueous sodium hydroxide solution dropwise to adjust the pH value to 8-9, then add 14.5g of tetrabutylammonium bromide, and heat up To 70 DEG C, begin to drop slowly 145ml diethyl sulfate (completely dropwise in about 4 hours), add 20% sodium hydroxide solution at any time during the dropping process, keep the pH of the reaction solution at 9-10, and keep The temperature of the reaction solution was 75-80° C., and the dropwise addition was completed (thin-layer chromatography showed that the raw material disappeared).
[0049] The reaction solution was lowered to room temperature, concentrated hydrochloric acid was added dropwise to adjust the pH value of the reaction solution to 2-3, extracted twice with cyclohexane (400ml×2), the organic phases were combined, and 400ml of saturated sodium chloride and 400ml of distilled water were respectively use...
Embodiment 3
[0051] Dissolve 88g of o-ethoxyphenol in 176ml of 1,2-dibromoethane, add 10.27g of tetrabutylammonium bromide, raise the temperature to 75°C, then slowly add 640ml of 10% sodium hydroxide solution dropwise (about 6 hours dropwise), keep the pH value of the reaction solution between 9-10, dropwise is complete (thin-layer chromatography shows that the raw material disappears).
[0052] Separate the organic layer, wash the organic layer with 200ml of saturated sodium chloride solution and 200ml of water respectively, concentrate the organic layer to dryness, and recrystallize the crude product with 200mL of ethanol to obtain 2-(2-ethoxyphenoxy)ethyl Bromine 110g, the yield is 70.5%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More