Methanesulfonic acid imatinib tablet
A technology for ima mesylate and tinib tablets, which is applied in pill delivery, medical preparations of non-active ingredients, pharmaceutical formulas, etc., can solve the problems of high particle hardness, affecting production efficiency, solvent residue, etc., and achieve the appearance Good smoothness, rapid drug dissolution, and good powder fluidity
Inactive Publication Date: 2015-02-11
SHANDONG NEWTIME PHARMA
View PDF7 Cites 7 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Due to the physical and chemical properties of imatinib mesylate itself, dry granulation is used for the production of tablets. The viscosity of the raw materials increases after absorbing moisture, and the passage is blocked on the dry granulator. The production process needs to be frequently shut down for cleaning, which affects production efficiency. , and a large amount of dust is produced during production, which is not conducive to the health protection of producers. In addition, the particles made by dry method have high hardness and it is difficult to achieve rapid dissolution
[0005] CN101653424A discloses the process of wet granulation with water, but imatinib mesylate will produce greater viscosity when it encounters water, and it is easy to form agglomerates in granulation, which affects subsequent production
CN102349875A discloses a wet granulation method, using an organic solvent or an organic solvent solution with a volume concentration greater than 70% as the granulation solution, and the proportion of the water-insoluble filler in the tablet to the total weight of the tablet is less than 20%, but in the process A large amount of organic solvents are used, which will not only remain in the preparation, but also not conducive to labor protection
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0019] Prescription and preparation process of imatinib mesylate tablets
[0020]
[0021] Imatinib mesylate is passed through a 80-mesh sieve, mixed evenly with other auxiliary materials, and directly compressed into tablets.
Embodiment 2
[0023] Prescription and preparation process of imatinib mesylate tablets
[0024]
[0025] Imatinib mesylate is passed through a 80-mesh sieve, mixed evenly with other auxiliary materials, and directly compressed into tablets.
Embodiment 3
[0027] Prescription and preparation process of imatinib mesylate tablets
[0028]
[0029]
[0030] Imatinib mesylate is passed through a 80-mesh sieve, mixed evenly with other auxiliary materials, and directly compressed into tablets.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention discloses a methanesulfonic acid imatinib tablet. The methanesulfonic acid imatinib tablet comprises, by weight, 1 part of methanesulfonic acid imatinib, 0.1-1 part of gum acacia, 2-8 parts of fillers, 0.1-1 part of disintegrating agents and 0.05-1 part of lubricating agents. The components are evenly mixed to prepare the methanesulfonic acid imatinib tablet with a direct powder compression method. The direct tablet pressing technology is adopted in the methanesulfonic acid imatinib tablet; the powder mobility is better; the tablet weight is stable; the appearance is bright and clean; the medicine dissolution speed is high; the production cost is low; and the methanesulfonic acid imatinib tablet is suitable for industrialized mass production.
Description
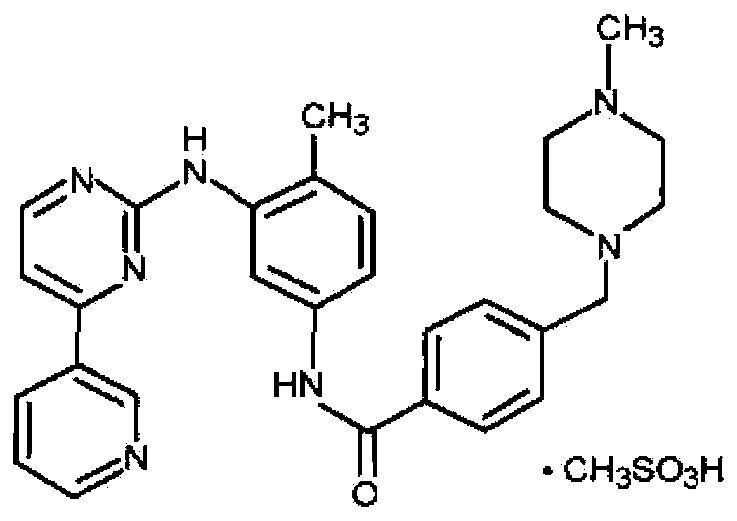
technical field [0001] The invention belongs to the technical field of pharmaceutical preparations, in particular to an imatinib mesylate tablet. Background technique [0002] The original research unit of imatinib mesylate tablets is Novartis, Switzerland, which was approved by the FDA in May 2001. The chemical name: 4-[(4-methyl-1-piperazine)methyl]-N-[4-methyl Base-3-[4-(3-pyridine)-2-pyrimidine]amino]phenyl]-aniline methanesulfonate, the structural formula is as follows: [0003] [0004] Imatinib mesylate is white to slightly yellow crystalline powder; tasteless and odorless. Soluble in water, 0.1mol / L hydrochloric acid, glacial acetic acid, slightly soluble in methanol, very slightly soluble in ethanol, almost insoluble in 0.1mol / L sodium hydroxide solution, acetonitrile. Due to the physical and chemical properties of imatinib mesylate itself, dry granulation is used for the production of tablets. The viscosity of the raw materials increases after absorbing moistu...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): A61K9/20A61K31/506A61K47/30
Inventor 赵志全郝贵周蒋新利
Owner SHANDONG NEWTIME PHARMA