Method for separating and analyzing trelagliptin succinate and preparation related substances thereof
A technology for the separation and analysis of troxagliptin succinate, which is applied in the field of chemical analysis, can solve problems such as inaccurate integration, and achieve the effect of ensuring product quality and good elution and separation effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0044] Instrument: Agilent 1100 HPLC, VWD detector
[0045] Chromatographic column: Octadecyl bonded silica gel as filler (4.6mm×250mm, 5μm)
[0046] Mobile phase A: 0.02mol / L potassium dihydrogen phosphate buffer solution (containing 0.3% by volume of triethylamine), adjust the pH to 3.0 with phosphoric acid
[0047] Mobile Phase B: Acetonitrile
[0048] The gradient elution is shown in Table 1 below:
[0049] Table 1
[0050]
[0051] Flow rate: 1.0mL / min
[0052] Wavelength: 224nm
[0053] Column temperature: 30°C
[0054] Injection volume: 10μL
[0055] Diluent: mobile phase A
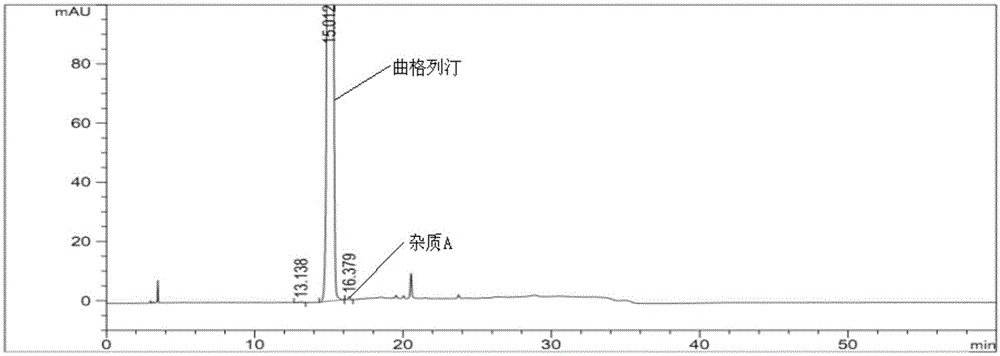
[0056]Experimental procedure: take trexagliptin tablets (the ratio of trexagliptin raw material to microcrystalline cellulose is 1:5), grind it into fine powder with a mortar, weigh the fine powder, and prepare each mL of koji with mobile phase A Gliptin 500μg solution was then analyzed under the above-mentioned gradient elution conditions, and the obtained spectra were as follows figu...
Embodiment 2
[0059] Instrument: Agilent 1100 HPLC, VWD detector
[0060] Chromatographic column: Octadecyl bonded silica gel as filler (4.6mm×250mm, 5μm)
[0061] Mobile phase A: 0.02mol / L sodium dihydrogen phosphate buffer solution (containing 0.3% by volume of triethylamine), adjust pH to 3.0 with phosphoric acid
[0062] Mobile Phase B: Acetonitrile
[0063] The gradient elution is shown in Table 2 below:
[0064] Table 2
[0065]
[0066]
[0067] Flow rate: 1.0mL / min
[0068] Wavelength: 224nm
[0069] Column temperature: 30°C
[0070] Injection volume: 10μL
[0071] Diluent: mobile phase A
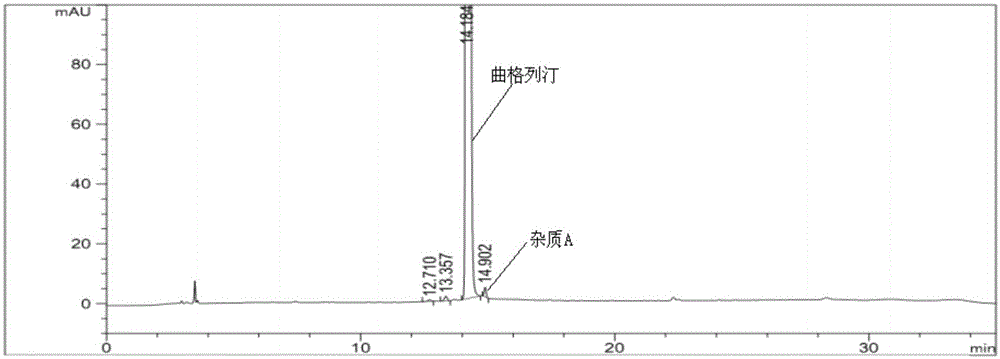
[0072] Experimental procedure: take trexagliptin tablets (the ratio of trexagliptin raw material to microcrystalline cellulose is 1:5), grind it into fine powder with a mortar, weigh the fine powder, and prepare each mL of koji with mobile phase A Gliptin 500μg solution was then analyzed under the above-mentioned gradient elution conditions, and the obtained spectra were as follows ...
Embodiment 3
[0075] Instrument: Agilent 1100 HPLC, VWD detector
[0076] Chromatographic column: Octadecyl bonded silica gel as filler (4.6mm×250mm, 5μm)
[0077] Mobile phase A: 0.02mol / L potassium dihydrogen phosphate buffer solution (containing 0.3% by volume of triethylamine), adjust the pH to 3.0 with phosphoric acid
[0078] Mobile Phase B: Acetonitrile
[0079] The gradient elution is shown in Table 3 below:
[0080] table 3
[0081]
[0082] Flow rate: 1.0mL / min
[0083] Wavelength: 224nm
[0084] Column temperature: 30°C
[0085] Injection volume: 10μL
[0086] Diluent: mobile phase A
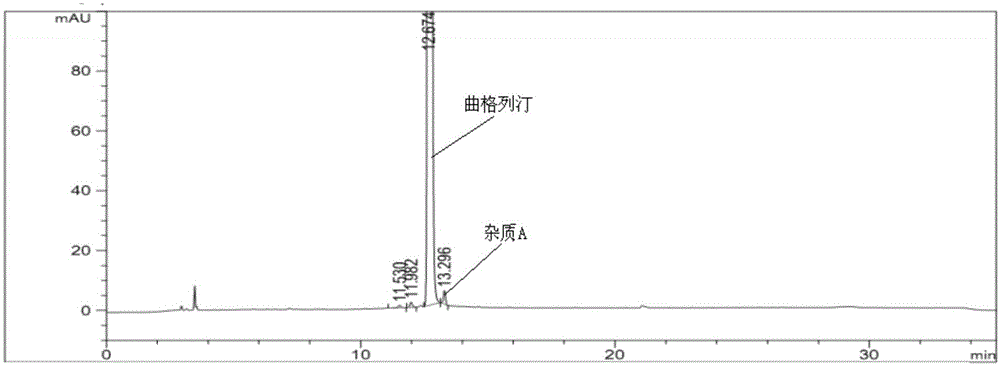
[0087] Experimental procedure: take trexagliptin tablets (the ratio of trexagliptin raw material to microcrystalline cellulose is 1:5), grind it into fine powder with a mortar, weigh the fine powder, and prepare each mL of koji with mobile phase A Gliptin 500μg solution was then analyzed under the above-mentioned gradient elution conditions, and the obtained spectra were as follows ima...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Concentration | aaaaa | aaaaa |
| Wavelength | aaaaa | aaaaa |
| Wavelength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More